
LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
1 | Page
v.9 April 2023
Table of Contents
Point of Care Testing - Reporting Requirements ........................................................................................... 2
Reporting Requirements .............................................................................................................................. 2
Manually Adding a Test Result for a New Individual ..................................................................................... 3
Setting Default POC Devices ......................................................................................................................... 5
Multiplex POC Devices ................................................................................................................................. 6
Adding Residents ......................................................................................................................................... 7
Adding Staff Demographic Data ................................................................................................................... 9
Uploading POC Test Results ........................................................................................................................ 12
Reporting Results for Individuals from Canada or Mexico............................................................................ 14
Reporting Results for Visitors ...................................................................................................................... 15
CMS Qualifications ...................................................................................................................................... 15
Clinical Laboratory Improvement Amendments (CLIA) Number ................................................................... 15
Secure Access Management Services (SAMS) Level ..................................................................................... 15
Testing Requirements ................................................................................................................................. 16
Resident, Staff, or Visitor ID ........................................................................................................................ 16
Race and Ethnicity ....................................................................................................................................... 19
Determining if Test Results Saved ............................................................................................................... 20
Deleting a Resident ..................................................................................................................................... 22
Deleting a Staff or Visitor ............................................................................................................................ 24
Deleting Test Results ................................................................................................................................... 26
Missing Option to Report POC Test Results ................................................................................................. 26
Missing Option to Report Staff or Visitor POC Results ................................................................................. 27
Group Access to POC Data ........................................................................................................................... 29
Training ....................................................................................................................................................... 30
Miscellaneous ............................................................................................................................................. 30

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
2 | Page
v.9 April 2023
Point of Care Testing - Reporting Requirements
Reporting Requirements
Q. Is there a requirement for nursing homes to report POC test results to NHSN?
CMS-certified long-term care facilities may submit point-of-care SARS-CoV-2 testing data, including antigen
testing data, to CDC’s National Healthcare Safety Network (NHSN). This CDC- and CMS-preferred pathway to
submit data to CDC’s NHSN applies only to CMS-certified long-term care facilities. Test data submitted to NHSN
will be reported to appropriate state and local health departments using standard electronic laboratory
messages. Other types of LTC facilities may also report testing data in NHSN for self-tracking or to fulfill state or
local reporting requirements, if any. While NHSN is the CDC- and CMS-preferred pathway, Medicare and
Medicaid-certified LTC facilities may submit data through the other mechanisms described in the Current
Methods of Submission section of HHS Laboratory Reporting Guidance
to meet the reporting requirements.
Q. Am I required to report COVID-19 test results for my facility?
Information regarding the requirement to report test results is located at
https://www.hhs.gov/sites/default/files/covid-19-laboratory-data-reporting-guidance.pdf
CMS is the regulatory body involved in enforcement of testing, not CDC. Further information can be found at:
https://www.cms.gov/newsroom/press-releases/cms-posts-guidance-implementing-new-testing-requirements-
nursing-homes-and-labs
CDC provides a pathway by which test results may be reported, but it is neither an enforcement agency nor the
agency that mandates testing or reporting.
Q. Must negative results also be reported?
No. Effective April 4, 2022, entities conducting all other SARS-COV-2 testing (e.g., testing conducted in a setting
operating under a CLIA certificate of waiver, non-NAAT testing conducted in a facility certified under CLIA to
perform moderate- or high-complexity tests) except antibody and self-administered testing, must report
positive test results. Reporting of negative results, either individual test results or in aggregate, is optional.
This includes rapid testing conducted in many settings (e.g., screening testing at schools, correctional facilities,
employee testing programs, long-term care facilities, and point-of-care testing performed in pharmacies,
medical provider offices, and drive-through testing sites). Negative result reporting may still be required by
applicable state or local law, and entities should check with the applicable state, territorial, local, and Tribal
(STLT) jurisdiction for specific reporting requirements.
https://www.hhs.gov/sites/default/files/covid-19-
laboratory-data-reporting-guidance.pdf
Q. Do my facility’s POC test results reported to NHSN get sent to my state health department?
POC test results for SARS CoV-2 entered into NHSN are sent within 15 minutes of saving in NHSN, to an
electronic platform at the Association for Public Health Laboratories via a secure messaging format. From there,
within another 2 hours, they are sent to the appropriate state health department and to the Centers for Disease
Control and the U.S. Department of Health and Human Services. Non-SARS CoV-2 test results are sent on the
same schedule but sent based on preferences provided by state health departments and only to those health

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
3 | Page
v.9 April 2023
departments with which NHSN has a Data Use Agreement (DUA). DUA’s specify which data is shared and how
such data may be used. If you are interested in whether your state health or local department has a DUA with
NHSN, please email, NHSNDUA@cdc.gov. See Multiplex POC Devices
for more information about non-SARS-CoV-
2 test results reporting via the NHSN POC Tool.
Q. What if I’m already reporting POC test results to my state or local health department?
If your state or local health department has already developed a process by which facilities report these data,
please contact those public health authorities before making any changes to the reporting process.
Q. Can I report my test results via the pharmacy or laboratory?
Point of care testing is defined by the American College of Pathologists as, “testing that is performed near or at
the site of a patient with the result leading to a possible change in the care of the patient.” Therefore, if your
facility is a CMS-certified long-term care facility, and it is performing POC testing, the facility is required to report
the POC test results, not the laboratory.
Alternatively, if your facility simply collects a specimen from the individual in the nursing home, then transports
the specimen to the laboratory for testing, the laboratory is required to report the results by way of one of the
options for reporting included in the Reporting Guidance cited above.
Only results for COVID-19 from point of care (POC) devices in CMS-certified long-term care facilities may be
reported to NHSN via the POC Test Reporting Tool.
If all specimens for COVID-19 from your facility are being sent to a laboratory for processing and resulting, then
those will be reported via the laboratory, to the state or local health department.
Q. How are laboratory testing results for COVID-19 reported?
Laboratories have other mechanisms by which they routinely report Notifiable Diseases, which correlate to
other reporting pathways in the Reporting Guidance
https://www.cms.gov/newsroom/press-releases/cms-
posts-guidance-implementing-new-testing-requirements-nursing-homes-and-labs. Therefore, they will report
any results for tests performed in their laboratories, in that manner.
Manually Adding a Test Result for a New Individual
Q. How do I manually add a test result for a new individual?
1. Choose POC Test Result Reporting from the COVID-19 option in the left navigation bar.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
4 | Page
v.9 April 2023
2. Choose the type of individual tested from the drop-down menu, either a Resident or
Staff/Volunteer/Contractor and then enter the demographic data for the individual.
3. Create a unique ID for the individual. If that ID has not been used before, you will get a message instructing
that you can go ahead and directly enter the new individual’s information.
4. Click Ok and then enter the required demographic data above the POC Test Results section. Then click on Add
Test Result.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
5 | Page
v.9 April 2023
5. Proceed with adding the test results on the next screen. You may set up defaults for the POC device used in
your facility and the ordering physician, by using the Set as Default options. For the POC device, simply click on
the device of choice from the drop-down list and then choose Save as Default. This device will then
automatically load as the default device for each test result but can be changed if needed. You may also set up
more than one default device if your facility is using more than one POC device. For more guidance, see
Setting
Default POC Devices. Likewise, clicking on the blue Physician box will allow one to set up a default Physician.
Once all test results are recorded, choose Save. The test result has been recorded.
Setting Default POC Devices
Q. How can I set more than one default POC device so that I don’t have to scroll through so many devices to
switch between devices when reporting results?
If your facility is using more than a single POC test device for testing residents and staff, it may be helpful to list
more than one default POC device in the POC Test Results section. This can save valuable person-time in
scrolling through a lengthy POC device list. You may save a primary default device and multiple additional
defaults.
1. After Selecting “Add Test Result” select “Choose Default” from the POC Test Result page. Scroll down and
locate the primary default POC device of choice and click on it.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
6 | Page
v.9 April 2023
2. Next, select one or more additional devices as defaults from the drop- down menu of all devices labeled
“Select Additional Defaults”. Note, if additional previous defaults have been set up, and they are no longer
desired, first choose “Uncheck all” before making your selections, and when completed, choose “Save as
default”. Alternatively, you may uncheck individual previously selected default devices simply by clicking on
already checked boxes. The checkmark will disappear.
Multiplex POC Devices
Q. I heard that we could report more than just COVID-19 testing now through the POC Tool. Is that true?
Yes. Any test result that is provided by a POC test device which is included in the NHSN COVID-19 POC Test
Result Reporting Tool, can now be reported in the same manner as SARS-CoV-2 test results to the NHSN.
LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
7 | Page
v.9 April 2023
Reporting these test results is optional, with the exception of the result(s) of the SARS-CoV-2 test(s), which are
required and must be reported in order to save the NHSN event.
Adding Residents
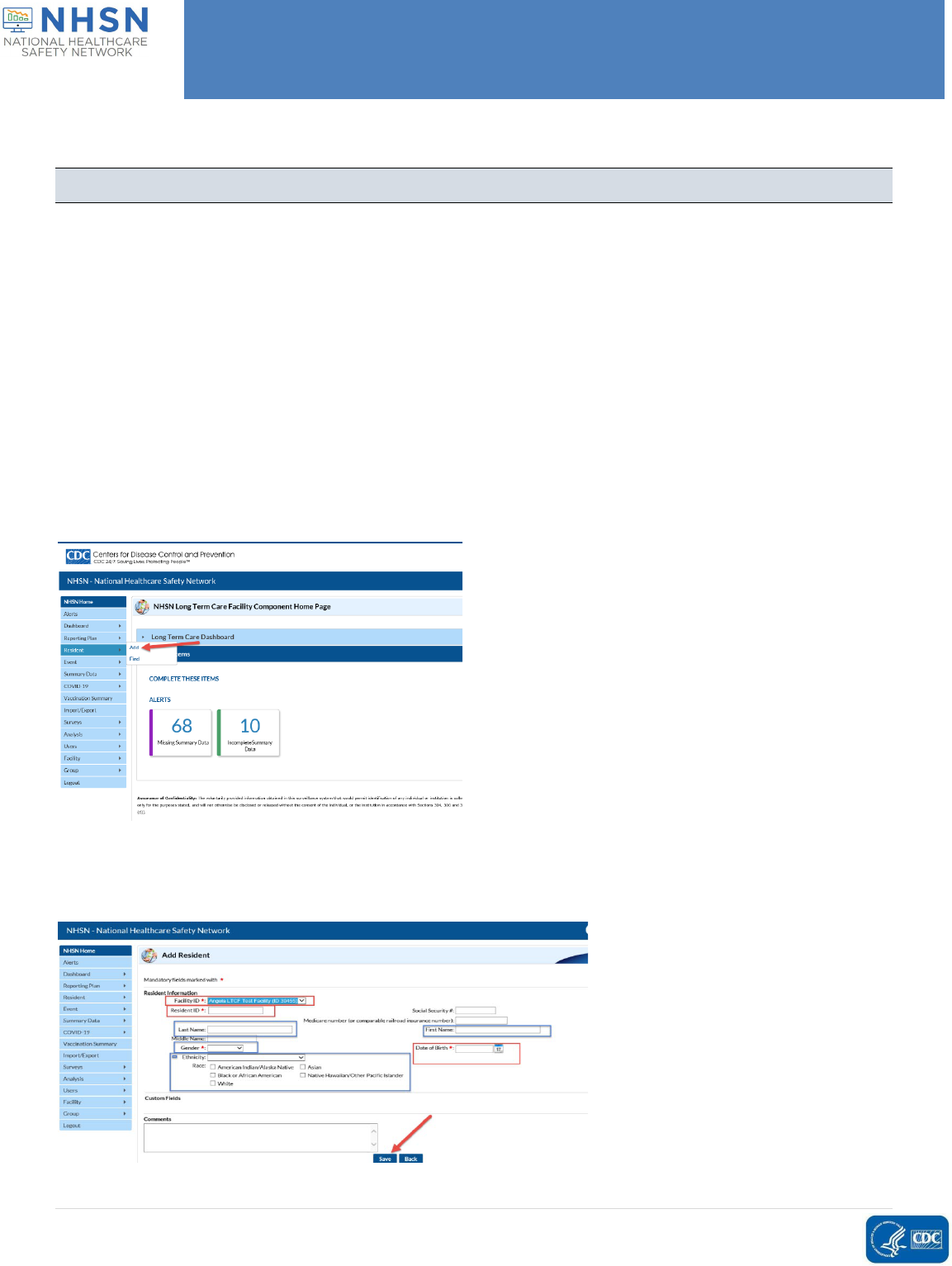
Q. How do I add a resident to the POC test reporting tool?
You may either batch upload demographic data for residents, or you may add the information individually for
each resident. See the question “How can I batch upload demographic information for residents in my
facility?” below for instructions on how to do this.
To individually add residents to the tool prior to inputting data, log into NHSN, find the Resident tab from the
left-hand side, hover over the tab and then click add. On the next page, complete not only the data fields with
red asterisks, (in red boxes below), which are generically required for ANY NHSN data entry, but also the data
fields in blue boxes which are required for Point of Care Test Reporting Tool data entry. Then hit Save. Enter
additional patients the same way.
Please refer to the screen shots below to better assist you.
You can go in and add Residents prior to testing.
On the next page, complete not only the data fields with red asterisks, (in red boxes below), which are
generically required for ANY NHSN data entry, but also the data fields in blue boxes which are required for
Point of Care Test Reporting Tool data entry. Then hit Save. Enter additional patients the same way.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
8 | Page
v.9 April 2023
Q. How can I batch upload demographic information for residents in my facility?
You may batch upload a Comma Separated Value (CSV) file of residents’ demographic data in the following
manner. Please see screen shots below to better assist you.
1. Access the option to import the data via the Import/Export option on the blue navigation panel, and then
Choose CSV Residents for the import type.
2. On the Import/Export Data screen, select the Choose File button then navigate to the location at which the
CSV file was saved.
3. Navigate to, and select, the file which you would like to upload.
4. Once back at the Import/Export Data screen, choose Submit.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
9 | Page
v.9 April 2023
5. If there is any data which is missing or does not fit the required format, those lines will be listed under the
“Bad Data” heading. Edit or delete each of the lines of data as needed.
6. The template for the CSV file and the File Layout document can be located under the Importing Data
Resources heading and the Point of Care Upload Resources bullet at
https://www.cdc.gov/nhsn/ltc/covid19/index.html
Adding Staff Demographic Data
Q. How do I pre-load Staff demographic data for the Point of Care Test Reporting Tool?
There are 2 ways to upload Staff demographic information. One is an upload via a Comma-separated Value
(CSV) format, and the other is a manual upload of individual staff’s demographic data one-by one. Please see
the guidance provided for each that follows.
Q. How do I manually enter Staff demographic data for the Point of Care Test Reporting Tool?
Please see below the screen shots of this process.
1. Choose POC Test Result Reporting from the COVID-19 option in the left navigation bar.
2. Indicate that the type of individual tested is a Staff/Volunteer/Contractor and then enter the demographic
data for the individual. Once the data is complete, choose Save Staff/Volunteer/Contractor Demographics.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
10 | Page
v.9 April 2023
3. You will encounter an “Alert” message that confirms that the demographic data has been successfully saved.
You will be given the choice to choose “Ok” if you are finished entering demographic data for
Staff/Volunteer/Contractors, or you may alternatively choose “New” if you want to enter demographic data for
another Staff/Volunteer/Contractor.
4. When POC test results need to be entered for the individual, go into the POC Test Result Reporting Screen,
enter the individual’s Staff ID and then click outside the box. Their demographic data will appear, and you can
choose Add Test Result near the bottom to add their POC test results.
5. The individual also appears in the list provided when you choose “Find Resident/Staff/Visitor”.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
11 | Page
v.9 April 2023
Q. How do I upload Staff demographic data for the Point of Care Test Reporting Tool?
You may batch upload a Comma Separated Value (CSV) file of staffs’ demographic data in the following manner.
Please see screen shots below to better assist you.
1. Access the option to import the data via the Import/Export option on the blue navigation panel, and then
Choose CSV Staff for the import type.
2. On the Import/Export Data screen, select the Choose File button then navigate to the location at which the
CSV file was saved.
3. Select the file which you would like to upload.
4. Once back at the Import/Export Data screen, choose “Submit”.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
12 | Page
v.9 April 2023
5. If there is any data which is missing or does not fit the required format, those lines will be listed under the
“Bad Data” heading. Edit or delete each of the lines of data as needed.
6. The template for the CSV file and the File Layout document can be located under the Importing Data
Resources heading and the Point of Care Upload Resources bullet at
https://www.cdc.gov/nhsn/ltc/covid19/index.html
Uploading POC Test Results
Q. Can I upload POC test results to NHSN?
Yes. You can upload POC test results via a comma-separated value (CSV) file. To do so, save the data in a CSV
file format that is in accordance with the File Layout for CSV Import LTCF COVID-19 POC Test Results found in
the Importing Data Resources section at this location:
https://www.cdc.gov/COVID-19 Module
LTCF/ltc/covid19/index.html
1. Prepare your file

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
13 | Page
v.9 April 2023
2. Verify the OrgID and CLIA ID (Facility Information) numbers match the file(s) to be uploaded.
3. Select COVID-19 on navigation bar
4. Select POC test Result Reporting
5. Scroll to bottom of page to select “Upload CSV”
6. Upload CSV File pop-up window displays
7. Select “Browse”

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
14 | Page
v.9 April 2023
8. Select the CSV file which you had previously prepared to Upload
9. Click “Upload CSV”
10. If the file has no errors, you will get an upload success message.
11. Verify record is uploaded:
a. On POC Test Result Reporting:
b. Select Find Resident/Staff/Visitor… tab, top right of page
c. Enter the ID you’re searching for in the ID field
d. Verify the record is correct.
12. If there are errors in the data which you are uploading to NHSN, you will encounter the CSV Upload Errors
screen. The line(s) of data containing error(s) will be identified and the field(s) containing the error(s) indicated.
It is suggested that you print a copy of the errors so that you can correct them. To do so, use the Print Screen
(prt sc) key on your keyboard to capture all the records that have errors. Note, you can adjust the numbers of
lines that are shown on the screen, and you may need to print more than one screen. Once this is completed
select “Cancel”, return to the data file, correct all incorrect data, and then restart the CSV upload process. Once
all errors are corrected, the file will successfully upload.
If an incorrect file type or blank .csv file is uploaded you will receive the same error message, but no errors will
display. You should upload a .csv file and provide a .csv file with data.
Reporting Results for Individuals from Canada or Mexico
Q. If we have facility staff who reside in Canada or Mexico and cross the border to work in our facility, how do
we report those results?
POC results for staff members who live in Canada or Mexico can be added to the NHSN application in the same
way that those who live in the U.S. are added. You will simply select Canada or Mexico from the drop-down
menu for the data field Country and then the phone number and zip code must meet the required format for
the selected country. The data field “County” will not be a required data field for those living in Canada or
Mexico.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
15 | Page
v.9 April 2023
Reporting Results for Visitors
Q. How do I report POC Test Results for visitors?
Access the POC Test Result Reporting page as is done for Resident and Staff. Choose “Visitor” from the drop-
down menu for Type of Individual Tested. Then proceed to complete the required data fields and save the data.
Q. Must I report POC test results for visitors?
Facilities that perform POC testing are required to report results for all POC testing that is performed within the
facility. If NHSN is the method by which POC test results are reported, then POC test results for visitors must be
reported to NHSN.
CMS Qualifications
Q. What is the definition of CMS-certified?
CMS-certified long-term care facilities consist of (1) skilled nursing facilities certified to participate in Medicare,
(2) nursing facilities certified to participate in Medicaid, and (3) facilities certified to participate in both programs
as a dually participating facility. The “other” category is defined as long term care facilities that have not been
certified by CMS. More information is at:
https://www.cms.gov/Medicare/Provider-Enrollment-and-
Certification/CertificationandComplianc/NHs
Clinical Laboratory Improvement Amendments (CLIA) Number
Q. How do I update or enter my facility’s CLIA number?
Only an NHSN facility administrator or an NHSN user within the facility who has administrative rights, can add or
edit the CLIA number. To do so, go to the Facility option in the blue navigation bar on the left of the screen when
you are logged into your facility. From that option, choose Facility Info. On that screen the CLIA number can
either be updated or added. Be sure to choose “Update” at the bottom of the page after making your changes to
ensure all changes are saved.
Secure Access Management Services (SAMS) Level
Q. How do I level up/proof up from SAMS level 1 to level 3 so that I can report POC data?
To assist our LTC partners in the NHSN community who are tasked with reporting patient-level Point of Care
COVID-19 testing results in addition to other facility-level COVID-19 reporting, we proactively began issuing CDC
SAMS Level 3 account invites to existing Level 1 user accounts. If your account has not been upgraded to a Level
3 account, which uses a secondary authentication Token, instead of a Level 1 account that just has a user ID and
password, then we recommend the following steps:
1. Please check your email backlog for an email from sams-no-[email protected]v
with a Level 3 proof up account
link and follow the link and associated instructions. The subject of the email is U.S. Centers for Disease
Control (CDC): SAMS Partner Portal - Identity Verification Request. Please ensure that your state ID match
your name and address provided in your already existing SAMS profile and please submit these documents
on the ID verification form found in the email from SAMS. Please ensure that your state ID match your name
and address provided on your SAMS profile

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
16 | Page
v.9 April 2023
2. If you do not have an email from step 1 then please contact the NHSN Help Desk at nhsn@cdc.gov
requesting an invite to secure Level 3 SAMS Clearance
3. Please note, if someone in your facility other than the NHSN Facility Administrator has retained a Level 3
SAMS clearance, either a Grid card Token or a mobile soft Token, this user can be granted administrative
rights and report on behalf of your facility. The NHSN Facility Administrator will need to grant permissions
to allow for patient-level data entry.
Testing Requirements
Q. Is testing for COVID-19 mandatory?
CMS is the regulatory body involved in enforcement, not CDC. Here is a site which can provide further
assistance:
https://www.cms.gov/newsroom/press-releases/cms-posts-guidance-implementing-new-testing-
requirements-nursing-homes-and-labs
The requirement to REPORT test results: https://www.hhs.gov/sites/default/files/covid-19-laboratory-data-
reporting-guidance.pdf
CDC provides a pathway by which test results may be reported, but it is neither an enforcement agency nor the
agency that mandates testing or reporting.
Resident, Staff, or Visitor ID
Q. Where can I find information about how to assign a Resident, Staff or Visitor ID#?
Please go to the following website:
https://www.cdc.gov/nhsn/ltc/covid19/index.html
Once there, scroll down the page to Data Collection Forms and Instructions and the last item under that heading
is "Point of Care Testing". The first document is the data collection tool for POC Test Reporting Tool, and the
second is the Table of Instructions for the data collection tool. That document provides guidance on completion
of the Resident Staff or Visitor ID#. IDs must be unique to the resident/staff/visitor. The system will not allow
two individuals to share the same ID. Birthdates are NOT recommended as ID numbers.
Q. How do I change/edit the Resident, Staff or Visitor ID?
1. Choose “POC Test Result Reporting” from the COVID-19 reporting option in the left navigation bar.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
17 | Page
v.9 April 2023
2. From the “POC Test Result Reporting” screen click “Find Resident/Staff/Visitor”.
3. Find the individual for whom you wish to edit their ID from the drop-down menu; then click on the individual
from the list provided and click “Select”.
4. Next, click “Edit Resident/Staff/Visitor”.
5. The fields will now appear white, indicating that they can be edited.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
18 | Page
v.9 April 2023
6. Once you have edited the ID field, you will receive a message stating, “Could not find Resident (Staff or
Visitor-depending on screen). If you would like to proceed with editing the ID, select OK. Otherwise, select
Cancel.” Select “OK”.
7. Next, click “Save Resident/Staff/Visitor”.
8. You will then be taken back to the “POC Test Result Reporting” screen where you can proceed with entering
POC test results.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
19 | Page
v.9 April 2023
9. Optional: The OLD ID will remain in the resident/staff/visitor list; you are able to delete the OLD
resident/staff/visitor if needed.
Note: The test results will no longer appear with the OLD resident/staff/visitor ID, test results will ONLY appear
with the new/edited resident/staff/visitor ID. Please see the sections titled, “Deleting a Resident” or “Deleting a
Staff or Visitor” for further instructions if you wish to proceed with this step.
Race and Ethnicity
Q. What if I do not know the race or ethnicity of my resident?
Collecting race and ethnicity is important for understanding trends in the COVID-19 pandemic and ensuring the
wellbeing of racial and ethnic minority groups. Because of this, race and ethnicity are U.S. Department of Health
and Human Services required data fields for POC Test Reporting. In basic terms, race describes physical traits,
and ethnicity refers to cultural identification. Race may also be identified as something you inherit
while ethnicity is something you learn. NHSN classifies race according to the 5 races included in the Office of
Management and Budget’s (OMB) issued the Revisions to the Standards for the Classification of Federal Data on
Race and Ethnicity https://obamawhitehouse.archives.gov/omb/fedreg_1997standards
• American Indian/Alaska Native
• Asian
• Black or African American
• Native Hawaiian/Other Pacific Islander
• White

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
20 | Page
v.9 April 2023
Please note that more than one race may be elected for an individual.
Likewise, ethnicity can be categorized as:
• Hispanic or Latino
• Not Hispanic or Latino
Hispanic or Latino is defined as a person of Cuban, Mexican, Puerto Rican, South or Central American, or other
Spanish culture or origin regardless of race. *
The resident should always be asked to identify their race and ethnicity. If the resident is not a good historian,
then check with a close family member. If, all good faith attempts to identify the race and/or ethnicity
information have failed, one of the following options may be chosen, as appropriate:
• Declined to respond
• Unknown
* https://www.census.gov/topics/population/hispanic-origin/about.html
Determining if Test Results Saved
Q. I am trying to add test results into NHSN but when I save the information, the test information is not
showing, just the resident/staff information. Is my data saved?
Once you hit "SAVE" you will not receive a confirmation. However, there are a few options you can perform to
check. If you see the button “I’m done, start a new POC test result” that is a good indicator that your data is
saved. You can also check if your data saved by searching for the individual, then looking to see if their test
results appear at the bottom of the page. You can also run a line list after generating datasets.
Please see below the screen shots of these options to better assist you.
1. You should see the test notification button (below).
2. Use the Find Resident/Staff option within the POC Test Reporting Tool, and then look for the test result
at the bottom.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
21 | Page
v.9 April 2023
3. Run a line list after generating datasets.
Step 1: Generate Dataset
Select> Analysis> Generate Data Set
Step 2: Generate Report
Select > Analysis > Reports

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
22 | Page
v.9 April 2023
Select Line Listing (Resident or Staff)
Select: Run Report
Deleting a Resident
(Note: Deleting a record is recommended only if the resident was accidentally added to the wrong facility; it
is NOT recommended for residents no longer living at the facility or who have died. Deleting the resident and
their record completely removes all evidence of any prior reporting of the data.)
Q. How do I delete a Resident?
To delete a Resident:
1. Choose Resident and then Find.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
23 | Page
v.9 April 2023
2. Locate the Resident.
3. Click the button next to the resident and then click the delete button at the top. If the resident has associated
Point of Care test results, you will be notified and you will need to go to the POC Test Results Reporting screen
and delete each of the POC test results before re-attempting to delete the Resident. Please also note that
deleting the Resident will also delete any associated healthcare-associated infections, for example, catheter-
associated urinary tract infections. NOTE: If you will need to report these test results for another individual (for
example you recorded these under the wrong Resident ID), make sure to record the results outside the
application BEFORE you delete them, so that you can enter them for the correct individual.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
24 | Page
v.9 April 2023
4. Once there are no point of care test results are associated with the resident, and you have repeated steps 1-3,
you will be provided a prompt to confirm you want to delete the Resident. Click Delete.
Deleting a Staff or Visitor
(Note: Deleting a record is recommended only if the staff/visitor was accidentally added to the wrong facility;
deleting the staff/visitor and their record completely removes all evidence of any prior reporting of the data.)
Q. How do I delete a staff person or visitor from the system?
The process for deleting a staff person or visitor is different than deleting a Resident.
1. Choose POC Test Result Reporting from the COVID-19 option in the left navigation bar.
2. From the POC Test Result Reporting screen click “Find Resident/Staff/Visitor” then click on the Type box and
choose Staff from the drop-down menu. Then click on the correct staff person from the list provided and click
Submit.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
25 | Page
v.9 April 2023
3. Click on Delete Staff. If there are associated POC test results, a message indicating the need to delete those
test results will pop up. Choose OK. Then click on the trash can icon under Delete in the POC Test Results
section. Once all results are deleted, once again click on the Delete Staff button and confirm deletion. NOTE: If
you will need to report these test results for another staff person (for example you recorded these under the
wrong Staff ID), make sure to record the record results somewhere BEFORE you delete them, so that you can
enter them for the correct individual.
NOTE: A visitor can be deleted from the system in the same way, with the following minor changes:
• In Step 2 above, click on the Type box and choose Visitor from the drop-down menu. Then click on the
correct visitor from the list provided and click Submit.
• In Step 3 above, click on “Delete Visitor”.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
26 | Page
v.9 April 2023
Deleting Test Results
Q. How do I delete test results?
Deleting test results is a simple process.
1. Simply find the individual for whom you wish to delete test results.
2. Then click on the trash can icon under “Delete” on the POC Test Result Reporting screen and then click on
“OK” to the prompt “Are you sure you want to delete this test result?”
Missing Option to Report POC Test Results
Q. What if I don’t see the POC Test Reporting Tool in the dropdown menu?
There are a few things you can check. Are you a new User? If so, have you been granted rights for viewing and
or entering, etc. POC data? Someone with administrative rights in your facility must grant you those rights
If your facility was previously a level 1 facility for SAMS and is currently in the works for proofing up to level 3,
please note the following. As a level 3 User, you must use your SAMS grid card as well as a different link to

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
27 | Page
v.9 April 2023
access the reporting. When you log in, do NOT use the “NHSN LTC Reporting” link that you were using previously
as a Level 1 user. Instead, use the “NHSN Reporting” option to log in. The POC option should then be available
for entry.
Missing Option to Report Staff or Visitor POC Results
Q. Why am I not seeing the option to report Point of Care (POC) test results for staff? How can I?
If an NHSN User has rights to add Staff POC test data, the option for staff will be listed in the drop- down menu
for Type of Individual Tested. However, for confidentiality reasons, NHSN has only defaulted the rights to enter
staff POC test result data to the NHSN Facility Administrator (FacAd). This means that other NHSN users will not
be able to add staff POC test data unless enabled by the NHSN FacAd. The NHSN FacAd can edit rights for other
Users in the system to enter the data should he/she choose to do so. The NHSN FacAd will need to do this
individually, for each person that they wish to have such rights.
Please see the screen shots below for steps for an NHSN FacAd to assign “add, enter or delete”, or “view” rights
to staff POC test data, to additional facility NHSN members.
1. Once in the NHSN application, choose Users, and then Find from the options on the blue navigation bar and
drop-down menu respectively.
2. Type in the last name of the individual who is already an NHSN User in the facility. Choose “Find”.
3. Once the user is located, on the View User screen, choose “Edit”.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
28 | Page
v.9 April 2023
4. On the Edit User screen, choose “Edit Rights”.
5. On the Edit User Rights Screen, check the appropriate box(es) indicating the desired rights for the NHSN
member, 1) Staff- Add, Edit, Delete, or 2) Staff – View. Then choose Save. Repeat the process for any others
facility NHSN members as needed.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
29 | Page
v.9 April 2023
Group Access to POC Data
Q. How can NHSN Groups access POC data?
There are 2 options for Groups within NHSN to access POC data in the POC Test Result Reporting Tool. One is to
view the data in line lists via the Analysis Option. The second option is to view the data within the POC Test
Result Reporting itself. Once in the NHSN system, choose the appropriate Group from the “Select facility/group”
option, then choose “All Facilities” from the “Select facility within the above group” option, and choose Submit.
1. From the left-hand navigation bar choose “COVID-19” and “POC Test Result Reporting”. This will open up the
data to the first of three levels- a listing of the data by facility and month.
2. In the screen shot below, the various categories for the months of data available for facility number 14529 (a
fictional facility) are shown. First there is an aggregation and statistics of Influenza virus tests performed in
March 2021. 4 tests were performed, all were reported as negative. In the same month, the facility reported 46
SARS CoV-2 antibody tests, of which 21, or 45.7% were positive, as well as 63 tests performed for SARS CoV-2
virus of which 22 or 34.9% were positive. Likewise, there are SARS CoV-2 test results reported for the facility for
April 2021.

LONG-TERM CARE FACILITY (LTCF)
COVID-19 Point of Care (POC) Test Result Reporting Tool
Frequently Asked Questions
30 | Page
v.9 April 2023
3. By clicking on a line of data in the Summary Level 1, the specific test data for that line is displayed in the
Summary Level 2, below. This screen shot displays both an IgG and IgM antibody results as well as other types
of test results.
Training
Q. Where can I get guidance on using the POC Test Reporting Pathway?
A recording of previously offered trainings which are still current are posted to the NHSN website under the
heading “Archived Training” at this site: https://www.cdc.gov/nhsn/ltc/covid19/index.html
Miscellaneous
Q. How do I order POC test devices?
Please contact the Office for the Assistance Secretary of Health (OASH). Their email address for this purpose is:
NHTesting@hhs.gov
.
Q. How do I use the COVID-19 test kits?
Unfortunately, CDC does not handle the use and or guidelines of the COVID-19 tests, however the e-mail
address listed below may be able to offer advice. NHTesting@hhs.gov
. Device manufacturers should also be able
to provide guidance.
