
EN EN
EUROPEAN
COMMISSION
Brussels, 22.8.2022
C(2022) 5938 final
GUIDELINES
The Rules Governing Medicinal Products in the European Union
Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for
Human and Veterinary Use

1
The Rules Governing Medicinal Products in the European Union
Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for
Human and Veterinary Use
Annex 1
Manufacture of Sterile Medicinal Products
Legal context for publishing the detailed guidelines: Article 47 of Directive 2001/83/EC
on the Community code relating to medicinal products for human use and Regulation 2019/6
on the Community code relating to veterinary medicinal products. This document provides
technical guidance on the principles and guidelines of good manufacturing practice (GMP)
for medicinal products as laid down in Commission Directive (EU) 2017/1572 for medicinal
products for human use, Directive 91/412/EEC for veterinary use, and Commission
Delegated Regulation (EU) 2017/1569 for investigational medicinal products for human use
and arrangements for inspections supplementing Regulation (EU) No 536/2014 on clinical
trials.
This Annex is intended to assist national authorities in the application of the EU legislation.
Only the Court of Justice of the European Union is competent to authoritatively interpret
Union law.
Status of the document: Revision of the 2007 version of Annex 1.
Document History
Previous version dated 30 May 2003, in operation since
September 2003
Revision to align classification table of clean rooms, to
includeguidance on media simultations, bioburden
monitoring and capping of vials
November 2005 to December 2007
Date for coming into operation and superseding
01 March 2009/01 March 2010
Note: Provisions on capping of vials were
implemented on 01 March 2010.
Reasons for changes: The GMP/GDP Inspectors Working Group and the PIC/S Committee
jointly recommend that the current version of annex 1, on the manufacture of sterile
medicinal products, is revised to reflect changes in regulatory and manufacturing
environments. The new guideline should clarify how manufacturers can take advantage of
new possibilities deriving from the application of an enhanced process understanding by
using innovative tools as described in the ICH Q9 and Q10 guidelines.
The revision of Annex 1 should also take into account related changes in other GMP chapters
and annexes as well as in other regulatory documents. The revised guideline will seek to
remove ambiguity and inconsistencies and will take account of advances in technologies.
Deadline for coming into operation:
- 25 August 2023 : one year from the date of publication in Eudralex Volume 4
- 25 August 2024 : two years from the date of publication in Eudralex Volume 4 for
point 8.123

2
Document map
Section Number
General overview
1. Scope
Includes additional areas (other than sterile products) where the
general principles of the annex can be applied.
2. Principle
General principles as applied to the manufacture of sterile
products.
3. Pharmaceutical Quality
System (PQS)
Highlights the specific requirements of the PQS when applied
to sterile products.
4. Premises
General guidance regarding the specific needs for premises
design and also guidance on the qualification of premises
including the use of Barrier Technology.
5. Equipment
General guidance on the design and operation of equipment.
6. Utilities
Guidance regarding the special requirements of utilities such as
water, gas and vacuum.
7. Personnel
Guidance on the requirements for specific training, knowledge
and skills. Also gives guidance regarding the qualification of
personnel.
8. Production and specific
technologies
Guidance on the approaches to be taken regarding aseptic and
terminal sterilization processes. Guidance on the approaches to
sterilization of products, equipment and packaging
components. Also guidance on different technologies such as
lyophilization and Form-Fill-Seal where specific requirements
apply.
9. Environmental and process
monitoring
This section differs from guidance given in section 4 in that the
guidance here applies to ongoing routine monitoring regarding
the design of systems and setting of action limits alert levels
and reviewing trend data.
The section also gives guidance on the requirements of Aseptic
Process Simulations (APS).
10. Quality control (QC)
Guidance on some of the specific Quality Control requirements
relating to sterile products.
11. Glossary
Explanation of specific terminology.

3
1 Scope
The manufacture of sterile products covers a wide range of sterile product types (active substance,
excipient, primary packaging material and finished dosage form), packed sizes (single unit to multiple
units), processes (from highly automated systems to manual processes) and technologies (e.g.
biotechnology, classical small molecule manufacturing systems and closed systems). This Annex
provides general guidance that should be used in the design and control of facilities, equipment,
systems and procedures used for the manufacture of all sterile products applying the principles of
Quality Risk Management (QRM), to ensure that microbial, particulate and endotoxin/pyrogen
contamination is prevented in the final product.
QRM applies to this document in its entirety and will not, normally, be referred to in specific
paragraphs. Where specific limits or frequencies or ranges are specified, these should be considered as
a minimum requirement. They are stated due to historical regulatory experience of issues that have
been identified and have impacted the safety of patients.
The intent of the Annex is to provide guidance for the manufacture of sterile products. However,
some of the principles and guidance, such as contamination control strategy, design of premises,
cleanroom classification, qualification, validation, monitoring and personnel gowning, may be used to
support the manufacture of other products that are not intended to be sterile such as certain liquids,
creams, ointments and low bioburden biological intermediates, but where the control and reduction of
microbial, particulate and endotoxin/pyrogen contamination is considered important. Where a
manufacturer elects to apply guidance herein to non-sterile products, the manufacturer should clearly
document which principles have been applied and acknowledge that compliance with those principles
should be demonstrated.
2 Principle
2.1 The manufacture of sterile products is subject to special requirements in order to minimize risks of
microbial, particulate and endotoxin/pyrogen contamination. The following key areas should be
considered:
i. Facility, equipment and process should be appropriately designed, qualified and/or validated
and where applicable, subjected to ongoing verification according to the relevant sections of
the Good Manufacturing Practices (GMP) guidelines. The use of appropriate technologies
(e.g. Restricted Access Barriers Systems (RABS), isolators, robotic systems,
rapid/alternative methods and continuous monitoring systems) should be considered to
increase the protection of the product from potential extraneous sources of
endotoxin/pyrogen, particulate and microbial contamination such as personnel, materials
and the surrounding environment, and assist in the rapid detection of potential contaminants
in the environment and the product.
ii. Personnel should have adequate qualifications and experience, training and behaviour with a
specific focus on the principles involved in the protection of sterile product during the
manufacturing, packaging and distribution processes.
iii. Processes and monitoring systems for sterile product manufacture should be designed,
commissioned, qualified, monitored and regularly reviewed by personnel with appropriate
process, engineering and microbiological knowledge.
iv. Raw materials and packaging materials should be adequately controlled and tested to ensure
that level of bioburden and endotoxin/pyrogen are suitable for use.
4
2.2 Processes, equipment, facilities and manufacturing activities should be managed in accordance
with QRM principles to provide a proactive means of identifying, scientifically evaluating and
controlling potential risks to quality. Where alternative approaches are used, these should be
supported by appropriate rationale, risk assessment and mitigation, and should meet the intent of this
Annex.
In the first instance, QRM priorities should include appropriate design of the facility, equipment and
processes, followed by the implementation of well-designed procedures, and finally application of
monitoring systems as the element that demonstrates that the design and procedures have been
correctly implemented and continue to perform in line with expectations. Monitoring or testing alone
does not give assurance of sterility.
2.3 A Contamination Control Strategy (CCS) should be implemented across the facility in order to
define all critical control points and assess the effectiveness of all the controls (design, procedural,
technical and organisational) and monitoring measures employed to manage risks to medicinal
product quality and safety. The combined strategy of the CCS should establish robust assurance of
contamination prevention. The CCS should be actively reviewed and, where appropriate, updated and
should drive continual improvement of the manufacturing and control methods. Its effectiveness
should form part of the periodic management review. Where existing control systems are in place and
are appropriately managed, these may not require replacement but should be referenced in the CCS
and the associated interactions between systems should be understood.
2.4 Contamination control and steps taken to minimize the risk of contamination from microbial,
endotoxin/pyrogen and particle sources includes a series of interrelated events and measures. These
are typically assessed, controlled and monitored individually but their collective effectiveness should
be considered together.
2.5 The development of the CCS requires detailed technical and process knowledge. Potential sources
of contamination are attributable to microbial and cellular debris (e.g. pyrogen, endotoxin) as well as
particulate (e.g. glass and other visible and sub-visible particles).
Elements to be considered within a CCS should include (but are not limited to):
i. Design of both the plant and processes including the associated documentation.
ii. Premises and equipment.
iii. Personnel.
iv. Utilities.
v. Raw material controls – including in-process controls.
vi. Product containers and closures.
vii. Vendor approval – such as key component suppliers, sterilisation of components and single
use systems (SUS), and critical service providers.
viii. Management of outsourced activities and availability/transfer of critical information
between parties, e.g. contract sterilisation services.
ix. Process risk management.
x. Process validation.
xi. Validation of sterilisation processes.

5
xii. Preventative maintenance – maintaining equipment, utilities and premises (planned and
unplanned maintenance) to a standard that will ensure there is no additional risk of
contamination.
xiii. Cleaning and disinfection.
xiv. Monitoring systems - including an assessment of the feasibility of the introduction of
scientifically sound, alternative methods that optimize the detection of environmental
contamination.
xv. Prevention mechanisms – trend analysis, detailed investigation, root cause determination,
corrective and preventive actions (CAPA) and the need for comprehensive investigational
tools.
xvi. Continuous improvement based on information derived from the above.
2.6 The CCS should consider all aspects of contamination control with ongoing and periodic review
resulting in updates within the pharmaceutical quality system as appropriate. Changes to the systems
in place should be assessed for any impact on the CCS before and after implementation.
2.7 The manufacturer should take all steps and precautions necessary to assure the sterility of the
products manufactured within its facilities. Sole reliance for sterility or other quality aspects should
not be placed on any terminal process or finished product test.
3 Pharmaceutical Quality System (PQS)
3.1 The manufacture of sterile products is a complex activity that requires specific controls and
measures to ensure the quality of products manufactured. Accordingly, the manufacturer’s PQS
should encompass and address the specific requirements of sterile product manufacture and ensure
that all activities are effectively controlled so that the risk of microbial, particulate and
endotoxin/pyrogen contamination is minimized in sterile products. In addition to the PQS
requirements detailed in Chapter 1 of the GMP guidelines (Part I - Basic Requirements for Medicinal
Products), the PQS for sterile product manufacture should also ensure that:
i. An effective risk management system is integrated into all areas of the product life cycle with
the aim to minimize microbial contamination and to ensure the quality of sterile products
manufactured.
ii. The manufacturer has sufficient knowledge and expertise in relation to the products
manufactured and the equipment, engineering and manufacturing methods employed that
have an impact on product quality.
iii. Root cause analysis of procedural, process or equipment failure is performed in such a way
that the risk to product is correctly identified and understood so that suitable corrective and
preventive actions (CAPA) are implemented.
iv. Risk management is applied in the development and maintenance of the CCS, to identify,
assess, reduce/eliminate (where applicable) and control contamination risks. Risk
management should be documented and should include the rationale for decisions taken in
relation to risk reduction and acceptance of residual risk.
v. Senior management should effectively oversee the state of control throughout the facility and
product lifecycle. Risk management outcome should be reviewed regularly as part of the on-

6
going quality management, during change, in the event of a significant emerging problem,
and during the periodic product quality review.
vi. Processes associated with the finishing, storage and transport of sterile products should not
compromise the sterile product. Aspects that should be considered include: container
integrity, risks of contamination and avoidance of degradation by ensuring that products are
stored and maintained in accordance with the registered storage conditions.
vii. Persons responsible for the certification/release of sterile products have appropriate access to
manufacturing and quality information and possess adequate knowledge and experience in the
manufacture of sterile products and the associated critical quality attributes. This is in order to
allow such persons to determine if the sterile products have been manufactured in accordance
with the registered specifications and approved process and are of the required quality.
3.2 All non-conformities, such as sterility test failures, environmental monitoring excursions or
deviations from established procedures should be adequately investigated before certification/release
of the batch. The investigation should determine the potential impact upon process and product
quality and whether any other processes or batches are potentially impacted. The reason for including
or excluding a product or batch from the scope of the investigation should be clearly justified and
recorded.
4 Premises
4.1 The manufacture of sterile products should be carried out in appropriate cleanrooms, entry to
which should be through change rooms that act as airlocks for personnel and airlocks for equipment
and materials. Cleanrooms and change rooms should be maintained to an appropriate cleanliness
standard and supplied with air that has passed through filters of an appropriate efficiency. Controls
and monitoring should be scientifically justified and should effectively evaluate the state of
environmental conditions of cleanrooms, airlocks and pass-through hatches.
4.2 The various operations of component preparation, product preparation and filling should be
carried out with appropriate technical and operational separation measures within the cleanroom or
facility to prevent mix up and contamination.
4.3 Restricted Access Barrier Systems (RABS) or isolators are beneficial in assuring required
conditions and minimizing microbial contamination associated with direct human interventions in
the critical zone. Their use should be considered in the CCS. Any alternative approaches to the use
of RABS or isolators should be justified.
4.4 For the manufacture of sterile products, there are four grades of cleanroom/zone.
Grade A: The critical zone for high-risk operations (e.g. aseptic processing line, filling zone,
stopper bowl, open primary packaging or for making aseptic connections under the protection
of first air). Normally, such conditions are provided by a localised airflow protection, such as
unidirectional airflow workstations within RABS or isolators. The maintenance of
unidirectional airflow should be demonstrated and qualified across the whole of the grade A
area. Direct intervention (e.g. without the protection of barrier and glove port technology) into
the grade A area by operators should be minimized by premises, equipment, process and
procedural design.
Grade B: For aseptic preparation and filling, this is the background cleanroom for grade
A
(where it is not an isolator). Air pressure differences should be continuously monitored.
Cleanrooms of lower grade than grade B can be considered where isolator technology is used
(see paragraph 4.20 ).

7
Grade C and D: These are cleanrooms used for carrying out less critical stages in the
manufacture of aseptically filled sterile
products or as a background for isolators. They can
also be used for the preparation/filling of terminally sterilised products. (See section 8 for the
specific details on terminal sterilisation activities).
4.5 In cleanrooms and critical zones, all exposed surfaces should be smooth, impervious and
unbroken in order
to minimize the shedding or accumulation of particles or micro-organisms.
4.6 To reduce accumulation of dust and to facilitate cleaning there should be no recesses that are
difficult to clean effectively, therefore projecting ledges, shelves, cupboards and equipment should
be kept to a minimum. Doors should be designed to avoid recesses that cannot be cleaned. Sliding
doors may be undesirable for this reason.
4.7 Materials used in cleanrooms, both in the construction of the room and for items used within the
room, should be selected to minimize generation of particles and to permit the repeated application
of cleaning, disinfectant and sporicidal agents where used.
4.8 Ceilings should be designed and sealed to prevent contamination from the space above them.
4.9 Sinks and drains should be prohibited in the grade A and grade B areas. In other cleanrooms, air
breaks should be fitted between the machine or sink and the drains. Floor drains in lower grade
cleanrooms should be fitted with traps or water seals designed to prevent back flow and should be
regularly cleaned, disinfected and maintained.
4.10 The transfer of equipment and materials into and out of the cleanrooms and critical zones is one
of the greatest potential sources of contamination. Any activities with the potential to compromise
the cleanliness of cleanrooms or the critical zone should be assessed and if they cannot be
eliminated, appropriate controls should be implemented.
4.11 The transfer of materials, equipment, and components into the grade A or B areas should be
carried out via a unidirectional process. Where possible, items should be sterilised and passed into
these areas through double-ended sterilisers (e.g. through a double-door autoclave or depyrogenation
oven/tunnel) sealed into the wall. Where sterilisation upon transfer of the items is not possible, a
procedure which achieves the same objective of not introducing contamination should be validated
and implemented, (e.g. using an effective transfer disinfection process, rapid transfer systems for
isolators or, for gaseous or liquid materials, a bacteria-retentive filter). The removal of items from
the grade A and B areas (e.g. materials, waste, environmental samples) should be carried out via a
separate unidirectional process. If this is not possible, time-based separation of movement
(incoming/exiting material) by procedure should be considered and controls applied to avoid
potential contamination of incoming items.
4.12 Airlocks should be designed and used to provide physical separation and to minimize microbial
and particle contamination of the different areas and should be present for material and personnel
moving between different grades. Wherever possible, airlocks used for personnel movement should
be separated from those used for material movement. Where this is not practical, time-based
separation of movement (personnel/material) by procedure should be considered. Airlocks should be
flushed effectively with filtered air to ensure that the grade of the cleanroom is maintained. The final
stage of the airlock should, in the “at rest” state, be of the same cleanliness grade (viable and total
particle) as the cleanroom into which it leads. The use of separate change rooms for entering and
leaving the grade B area is desirable. Where this is not practical, time-based separation of activities
(ingress/egress) by procedure should be considered. Where the CCS indicates that the risk of
contamination is high, separate change rooms for entering and leaving production areas should be
used. Airlocks should be designed as follows:
8
i. Personnel airlocks: Areas of increasing cleanliness used for entry of personnel (e.g. from
the grade D area to the grade C area to the grade B area). In general hand washing facilities
should be provided only in the first stage of the changing room and not be present in
changing rooms directly accessing the grade B area.
ii. Material airlocks: used for materials and equipment transfer.
• Only materials and equipment that have been included on an approved list and
assessed during validation of the transfer process should be transferred into the grade
A or grade B areas via an airlock or pass-through hatches. Equipment and materials
(intended for use in the grade A area) should be protected when transiting through the
grade B area. Any unapproved items that require transfer should be pre-approved as
an exception. Appropriate risk assessment and mitigation measures should be applied
and recorded as per the manufacturer's CCS and should include a specific disinfection
and monitoring programme approved by quality assurance.
• Pass-through hatches should be designed to protect the higher-grade environment, for
example by effective flushing with an active filtered air supply.
• The movement of material or equipment from lower grade or unclassified area to
higher-grade clean areas should be subject to cleaning and disinfection commensurate
with the risk and in line with the CCS.
4.13 For pass-through hatches and airlocks (for material and personnel), the entry and exit doors
should not be opened simultaneously. For airlocks leading to the grade A and grade B areas, an
interlocking system should be used. For airlocks leading to grade C and D areas, a
visual and/or
audible warning system should be operated as a minimum. Where required to maintain area
segregation, a time delay between the closing and opening of interlocked doors should be
established.
4.14 Cleanrooms should be supplied with a filtered air supply that maintains a positive pressure
and/or an airflow relative to the background environment of a lower grade under all operational
conditions and should flush the area effectively. Adjacent rooms of different grades should have an
air pressure difference of a minimum of 10 Pascals (guidance value). Particular attention should be
paid to the protection of the critical zone. The recommendations regarding air supplies and pressures
may need to be modified where it is necessary to contain certain materials (e.g. pathogenic, highly
toxic or radioactive products or live viral or bacterial materials). The modification may include
positively or negatively pressurized airlocks that prevent the hazardous material from contaminating
surrounding areas. Decontamination of facilities (e.g. the cleanrooms and the heating, ventilation,
and air-conditioning (HVAC) systems) and the treatment of air leaving a clean area, may be
necessary for some operations. Where containment requires air to flow into a critical zone, the
source of the air should be from an area of the same or higher grade.
4.15 Airflow patterns within cleanrooms and zones should be visualised to demonstrate that there is
no ingress from lower grade to higher grade areas and that air does not travel from less clean areas
(such as the floor) or over operators or equipment that may transfer contamination to the higher
grade areas. Where unidirectional airflow is required, visualisation studies should be performed to
determine compliance, (see paragraphs 4.4 & 4.19). When filled, closed products are transferred to
an adjacent cleanroom of a lower grade via a small egress point, airflow visualization studies should
demonstrate that air does not ingress from the lower grade cleanrooms to the grade B area. Where air
movement is shown to be a contamination risk to the clean area or critical zone, corrective actions,
such as design improvement, should be implemented. Airflow pattern studies should be performed
both at rest and in operation (e.g. simulating operator interventions). Video recordings of the airflow
patterns should be retained. The outcome of the air visualisation studies should be documented and
9
considered when establishing the facility's environmental monitoring programme.
4.16 Indicators of air pressure differences should be fitted between cleanrooms and/or between
isolators and their background. Set points and the criticality of air pressure differences should be
considered within the CCS. Air pressure differences identified as critical should be continuously
monitored and recorded. A warning system should be in place to instantly indicate and warn
operators of any failure in the air supply or reduction of air pressure differences (below set limits for
those identified as critical). The warning signal should not be overridden without assessment and a
procedure should be available to outline the steps to be taken when a warning signal is given. Where
alarm delays are set, these should be assessed and justified within the CCS. Other air pressure
differences should be monitored and recorded at regular intervals.
4.17 Facilities should be designed to permit observation of production activities from outside the
grade A and B areas (e.g. through the provision of windows or remote cameras with a full view of
the area and processes to allow observation and supervision without entry). This requirement should
be considered when designing new facilities or during refurbishment of existing facilities.
Barrier Technologies
4.18 Isolators or RABS, which are different technologies, and the associated processes, should be
designed to provide protection through separation of the grade A environment from the environment
of the surrounding room. The hazards introduced from entry or removal of items during processing
should be minimized and supported by high capability transfer technologies or validated systems that
robustly prevent contamination and are appropriate for the respective technology.
4.19 The design of the technology and processes used should ensure appropriate conditions are
maintained in the critical zone to protect the exposed product during operations.
i. Isolators:
a. The design of open isolators should ensure grade A conditions with first air protection in
the critical zone and unidirectional airflow that sweeps over and away from exposed
products during processing.
b. The design of closed isolators should ensure grade A conditions with adequate protection
for exposed products during processing. Airflow may not be fully unidirectional in closed
isolators where simple operations are conducted. However, any turbulent airflow should
not increase risk of contamination of the exposed product. Where processing lines are
included in closed isolators, grade A conditions should be ensured with first air protection
in the critical zone and unidirectional airflow that sweeps over and away from exposed
products during processing
c. Negative pressure isolators should only be used when containment of the product is
considered essential (e.g. radiopharmaceutical products) and specialized risk control
measures should be applied to ensure the critical zone is not compromised.
ii. RABS:
The design of RABS should ensure grade A conditions with unidirectional airflow and first
air protection in the critical zone. A positive airflow from the critical zone to the supporting
background environment should be maintained.
10
4.20 The background environment for isolators or RABS should ensure the risk of transfer of
contamination is minimized.
i. Isolators:
a. The background environment for open isolators should generally correspond to a
minimum of grade C. The background for closed isolators should correspond to a
minimum of grade D. The decision on the background classification should be based on
risk assessment and justified in the CCS.
b. Key considerations when performing the risk assessment for the CCS of an isolator
should include (but are not limited to); the bio-decontamination programme, the extent
of automation, the impact of glove manipulations that may potentially compromise ‘first
air’ protection of critical process points, the impact of potential loss of barrier/glove
integrity, transfer mechanisms used and activities such as set-up or maintenance that
may require the doors to be opened prior to the final bio-decontamination of the isolator.
Where additional process risks are identified, a higher grade of background should be
considered unless appropriately justified in the CCS.
c. Airflow pattern studies should be performed at the interfaces of open isolators to
demonstrate the absence of air ingress.
ii. RABS:
The background environment for RABS used for aseptic processing should correspond to a
minimum of grade B and airflow pattern studies should be performed to demonstrate the
absence of air ingress during interventions, including door openings if applicable.
4.21 The materials used for glove systems (for both isolators and RABS), should be demonstrated to
have appropriate mechanical and chemical resistance. The frequency of glove replacement should be
defined within the CCS.
i. Isolators:
a. For isolators, leak testing of the glove system should be performed using a methodology
demonstrated to be suitable for the task and criticality. The testing should be performed at
defined intervals. Generally glove integrity testing should be performed at a minimum
frequency of the beginning and end of each batch or campaign. Additional glove integrity
testing may be necessary depending on the validated campaign length.
Glove integrity monitoring should include a visual inspection associated with each use
and following any manipulation that may affect the integrity of the system.
For manual aseptic processing activities where single unit or small batch sizes are
produced, the frequency of integrity verification may be based on other criteria, such as
the beginning and end of each manufacturing session.
b. Integrity / leak testing of isolator systems should be performed at defined intervals.
ii. RABS:
For RABS, gloves used in the grade A area should be sterilised before installation and
sterilised or effectively bio-decontaminated by a validated method prior to each
manufacturing campaign. If exposed to the background environment during operation,
disinfection using an approved methodology following each exposure should be completed.
Gloves should be visually examined with each use, and integrity testing should be
performed at periodic intervals.
11
4.22 Decontamination methods (cleaning and bio-decontamination, and where applicable inactivation
for biological materials) should be appropriately defined and controlled. The cleaning process prior to
the bio-decontamination step is essential; any residues that remain may inhibit the effectiveness of the
decontamination process. Evidence should also be available to demonstrate that the cleaning and bio-
decontamination agents used do not have adverse impact on the product produced within the RABS or
isolator.
i. For isolators
The bio-decontamination process of the interior should be automated, validated and
controlled within defined cycle parameters and should include a sporicidal agent in a
suitable form (e.g. gaseous or vaporized form). Gloves should be appropriately extended
with fingers separated to ensure contact with the agent. Methods used (cleaning and
sporicidal bio-decontamination) should render the interior surfaces and critical zone of the
isolator free from viable microorganisms.
ii. For RABS
The sporicidal disinfection should include the routine application of a sporicidal agent using
a method that has been validated and demonstrated to robustly include all areas of the
interior surfaces and ensure a suitable environment for aseptic processing.
Cleanroom and clean air equipment qualification
4.23 Cleanrooms and clean air equipment such as unidirectional airflow units (UDAFs), RABS and
isolators, used for the manufacture of sterile products, should be qualified according to the required
characteristics of the environment. Each manufacturing operation requires an appropriate
environmental cleanliness level in the operational state in order to minimize the risk of contamination
of the product or materials being handled. Appropriate cleanliness levels in the “at rest” and
“operational” states should be maintained.
4.24 Cleanrooms and clean air equipment should be qualified using methodology in accordance with
the requirements of Annex 15. Cleanroom qualification (including classification) should be clearly
differentiated from operational environmental monitoring.
4.25 Cleanroom and clean air equipment qualification is the overall process of assessing the level of
compliance of a classified cleanroom or clean air equipment with its intended use. As part of the
qualification requirements of Annex 15, the qualification of cleanrooms and clean air equipment
should include (where relevant to the design/operation of the installation):
i. Installed filter system leakage and integrity testing.
ii. Airflow tests - volume and velocity.
iii. Air pressure difference test.
iv. Airflow direction test and visualisation.
v. Microbial airborne and surface contamination.
vi. Temperature measurement test.
vii. Relative humidity test.

12
viii. Recovery test.
ix. Containment leak test.
Reference for the qualification of the cleanrooms and clean air equipment can be found in the ISO
14644 series of standards.
4.26 Cleanroom classification is part of the cleanroom qualification and is a method of assessing the
level of air cleanliness against a specification for a cleanroom or clean air equipment by measuring
the total particle concentration. Classification activities should be scheduled and performed in order
to avoid any impact on process or product quality. For example, initial classification should be
performed during simulated operations and reclassification performed during simulated operations or
during aseptic process simulation (APS).
4.27 For cleanroom classification, the total of particles equal to or greater than 0.5 and 5 µm should
be measured. This measurement should be performed both at rest and in simulated operations in
accordance with the limits specified in Table 1.
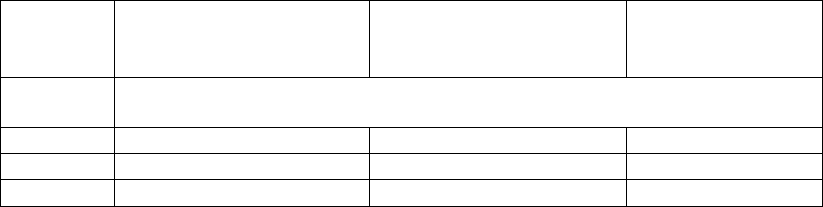
Table 1: Maximum permitted total particle concentration for classification
Grade
Maximum limits for total particle
≥ 0.5 µm/m
3
Maximum limits for total particle
≥ 5 µm/m
3
at rest
in operation
at rest
in operation
A
3 520
3 520
Not specified
(a)
Not specified
(a)
B
3 520
352 000
Not specified
(a)
2 930
C
352 000
3 520 000
2 930
29 300
D
3 520 000
Not
predetermined
(b)
29 300
Not
predetermined
(b)
(a)
Classification including 5µm particles may be considered where indicated by the CCS or
historical trends.
(b)
For grade D, in operation limits are not predetermined. The manufacturer should establish in
operation limits based on a risk assessment and routine data where applicable.
4.28 For classification of the cleanroom, the minimum number of sampling locations and their
positioning can be found in ISO 14644 Part 1. For the aseptic processing area and the background
environment (the grade A and grade B areas, respectively), additional sample locations should be
considered and all critical processing areas such as the point of fill and container closure feeder bowls
should be evaluated. Critical processing locations should be determined by documented risk
assessment and knowledge of the process and operations to be performed in the area.
4.29 Cleanroom classification should be carried out in the “at rest” and “in operation” states.
i. The definition of “at rest” state is the condition whereby the installation of all the utilities is
complete including any functioning HVAC, with the main manufacturing equipment installed
as specified but not operating and without personnel present in the room.
13
ii. The definition of “in operation” state is the condition where the installation of the cleanroom
is complete, the HVAC system fully operational, equipment installed and functioning in the
manufacturer’s defined operating mode with the maximum number of personnel present
performing or simulating routine operational work.
iii. The total particle limits given in Table 1 above for the “at rest” state should be achieved after
a “clean up” period on completion of operations and line clearance/cleaning activities. The
"clean up" period (guidance value of less than 20 minutes) should be determined during the
qualification of the rooms, documented and adhered to in procedures to reinstate a qualified
state of cleanliness if disrupted during operation.
4.30 The speed of air supplied by unidirectional airflow systems should be clearly justified in the
qualification protocol including the location for air speed measurement. Air speed should be designed,
measured and maintained to ensure that appropriate unidirectional air movement provides protection
of the product and open components at the working position (e.g. where high-risk operations occur
and where product and/or components are exposed). Unidirectional airflow systems should provide a
homogeneous air speed in a range of 0.36 – 0.54 m/s (guidance value) at the working position, unless
otherwise scientifically justified in the CCS. Airflow visualization studies should correlate with the air
speed measurement.
4.31 The microbial contamination level of the cleanrooms should be determined as part of the
cleanroom qualification. The number of sampling locations should be based on a documented risk
assessment and the results obtained from room classification, air visualization studies and knowledge
of the process and operations to be performed in the area. The maximum limits for microbial
contamination during qualification for each grade are given in Table 2. Qualification should include
both “at rest” and “in operation” states.
Table 2: Maximum permitted microbial contamination level during qualification
Grade
Air sample
CFU/m
3
Settle plates
(diameter 90 mm)
CFU/4 hours
(a)
Contact plates
(diameter 55
mm) CFU/plate
A
No growth
B
10
5
5
C
100
50
25
D
200
100
50
(a) Settle plates should be exposed for the duration of operations and changed as required after a maximum
of 4 hours. Exposure time should be based on recovery studies and should not allow desiccation of the
media used.
Note 1: All methods indicated for a specific grade in the table should be used for qualifying the area of
that specific grade. If one of the methods tabulated is not used, or alternative methods are used, the
approach taken should be appropriately justified.
Note 2: Limits are applied using CFU throughout the document. If different or new technologies are
used that present results in a manner different from CFU, the manufacturer should scientifically justify
the limits applied and where possible correlate them to CFU.
Note 3: For the qualification of personnel gowning, the limits given for contact plates and glove prints in
Table 6 should apply.
Note 4: Sampling methods should not pose a risk of contamination to the manufacturing operations.
14
4.32 The requalification of cleanrooms and clean air equipment should be carried out periodically
following defined procedures. The requalification should include at a minimum the following:
- Cleanroom classification (total particle concentration).
- Integrity test of final filters.
- Airflow volume measurement.
- Verification of air pressure difference between rooms.
- Air velocity test (Note: For grade B, C and D the air velocity test should be performed
according to a risk assessment documented as part of the CCS. However, it is required for filling
zones supplied with unidirectional airflow (e.g. when filling terminally sterilised products or
background to grade A and RABS). For grades with non-unidirectional airflow, a measurement
of recovery testing should replace velocity testing).
The maximum time interval for requalification of grade A & B areas, is 6 months.
The maximum time interval for requalification of grade C & D areas, is 12 months.
Appropriate requalification consisting of at least the above tests should also be carried out following
completion of remedial action implemented to rectify an out of compliance equipment or facility
condition or after changes to equipment, facility or processes as appropriate. The significance of a
change should be determined through the change management process. Examples of changes to be
considered include but are not limited to the following:
i. Interruption of air movement which affects the operation of the installation.
ii. Change in the design of the cleanroom or of the operational setting parameters of the
HVAC system.
iii. Special maintenance which affects the operation of the installation (e.g. change of final
filters).
Disinfection
4.33 The disinfection of cleanrooms is particularly important. They should be cleaned and disinfected
thoroughly in accordance with a written programme. For disinfection to be effective, prior cleaning to
remove surface contamination should be performed. Cleaning programmes should effectively remove
disinfectant residues. More than one type of disinfecting agent should be employed to ensure that
where they have different modes of action, their combined usage is effective against bacteria and
fungi. Disinfection should include the periodic use of a sporicidal agent. Monitoring should be
undertaken regularly in order to assess the effectiveness of the disinfection programme and to detect
changes in types of microbial flora (e.g. organisms resistant to the disinfection regime currently in
use).
4.34 The disinfection process should be validated. Validation studies should demonstrate the
suitability and effectiveness of disinfectants in the specific manner in which they are used and on the
type of surface material, or representative material if justified, and should support the in-use expiry
periods of prepared solutions.
4.35 Disinfectants and detergents used in grade A and grade B areas should be sterile prior to use.
Disinfectants used in grade C and D may also be required to be sterile where determined in the CCS.
Where the disinfectants and detergents are diluted / prepared by the sterile product manufacturer, this

15
should be done in a manner to prevent contamination and they should be monitored for microbial
contamination. Dilutions should be kept in previously cleaned containers (and sterilized where
applicable) and should only be stored for the defined period. If the disinfectants and detergents are
supplied “ready-made” then results from certificates of analysis or conformance can be accepted
subject to successful completion of the appropriate vendor qualification.
4.36 Where fumigation or vapour disinfection (e.g. Vapour-phase Hydrogen Peroxide) of cleanrooms
and associated surfaces are used, the effectiveness of any fumigation agent and dispersion system
should be understood and validated.
5 Equipment
5.1 A written, detailed description of the equipment design should be available (including process and
instrumentation diagrams as appropriate). This should form part of the initial qualification package
and be kept up to date.
5.2 Equipment monitoring requirements should be defined in “user requirements specifications”
during early stages of development, and confirmed during qualification. Process and equipment
alarm events should be acknowledged and evaluated for trends. The frequency at which alarms are
assessed should be based on their criticality (with critical alarms reviewed immediately).
5.3 As far as practicable, equipment, fittings and services should be designed and installed so that
operations, maintenance, and repairs can be performed outside the cleanroom. If maintenance has to
be performed in the cleanroom, and the required standards of cleanliness and/or asepsis cannot be
maintained, then precautions such as restricting access to the work area to specified personnel,
generation of clearly defined work protocols and maintenance procedures should be considered.
Additional cleaning, disinfection and environmental monitoring should also be considered. If
sterilisation of equipment is required, it should be carried out, wherever possible, after complete
reassembly.
5.4 The cleaning process should be validated to be able to:
i. Remove any residue or debris that would detrimentally impact the effectiveness of the
disinfecting agent used.
ii. Minimize chemical, microbial and particulate contamination of the product during the process
and prior to disinfection.
5.5 For aseptic processes, direct and indirect product contact parts should be sterilised. Direct
product contact parts are those that the product passes through, such as filling needles or pumps.
Indirect product contact parts are equipment parts that do not contact the product, but may come into
contact with other sterilised surfaces, the sterility of which is critical to the overall product sterility
(e.g. sterilised items such as stopper bowls and guides, and sterilised components).
5.6 All equipment such as sterilisers, air handling systems (including air filtration) and water
systems should be subject to qualification, monitoring and planned maintenance. Upon completion
of maintenance, their return to use should be approved.
5.7 Where unplanned maintenance of equipment critical to the sterility of the product is to be carried
out, an assessment of the potential impact to the sterility of the product should be performed and
recorded.
5.8 A conveyor belt should not pass through a partition between a grade A or B area and a
processing area of lower air cleanliness, unless the belt itself is continually sterilised (e.g. in a
sterilising tunnel).

16
5.9 Particle counters, including sampling tubing, should be qualified. The manufacturer’s
recommended specifications should be considered for tube diameter and bend radii. Tube length
should typically be no longer than 1m unless justified and the number of bends should be minimized.
Portable particle counters with a short length of sample tubing should be used for classification
purposes. Isokinetic sampling heads should be used in unidirectional airflow systems. They should
be oriented appropriately and positioned as close as possible to the critical location to ensure that
samples are representative.
6 Utilities
6.1 The nature and extent of controls applied to utility systems should be commensurate with the risk
to product quality associated with the utility. The impact should be determined via a risk assessment
and documented as part of the CCS.
6.2 In general, higher risk utilities are those that:
i. Directly contact product e.g. water for washing and rinsing, gases and steam for sterilisation.
ii. Contact materials that will ultimately become part of the product.
iii. Contact surfaces that come into contact with the product.
iv. Otherwise directly impact the product.
6.3 Utilities should be designed, installed, qualified, operated, maintained and monitored in a manner
to ensure that the utility system functions as expected.
6.4 Results for critical parameters and critical quality attributes of high risk utilities should be subject
to regular trend analysis to ensure that system capabilities remain appropriate.
6.5 Records of utility system installation should be maintained throughout the system’s life-cycle.
Such records should include current drawings and schematic diagrams, construction material lists and
system specifications. Typically, important information includes attributes such as:
i. Pipeline flow direction, slopes, diameter and length.
ii. Tank and vessel details.
iii. Valves, filters, drains, sampling and user points.
6.6 Pipes, ducts and other utilities should not be present in cleanrooms. If unavoidable, then they
should be installed so that they do not create recesses, unsealed openings and surfaces which are
difficult to clean. Installation should allow cleaning and disinfection of outer surface of the pipes.
Water systems
6.7 Water treatment plant and distribution systems should be designed, constructed, installed,
commissioned, qualified, monitored and maintained to prevent microbiological contamination and to
ensure a reliable source of water of an appropriate quality. Measures should be taken to minimize the
risk of presence of particulates, microbial contamination/proliferation and endotoxin/pyrogen (e.g.
sloping of piping to provide complete drainage and the avoidance of dead legs). Where filters are
included in the system, special attention should be given to their monitoring and maintenance. Water
produced should comply with the current monograph of the relevant Pharmacopeia.
6.8 Water systems should be qualified and validated to maintain the appropriate levels of physical,
17
chemical and microbial control, taking the effect of seasonal variation into account.
6.9 Water flow should remain turbulent through the pipes in water distribution systems to minimize
the risk of microbial adhesion, and subsequent biofilm formation. The flow rate should be established
during qualification and be routinely monitored.
6.10 Water for injections (WFI) should be produced from water meeting specifications that have
been defined during the qualification process, stored and distributed in a manner which minimizes
the risk of microbial growth (e.g. by constant circulation at a temperature above 70°C). WFI should
be produced by distillation or by a purification process that is equivalent to distillation. This may
include reverse osmosis coupled with other appropriate techniques such as electrodeionization
(EDI), ultrafiltration or nanofiltration.
6.11 Where WFI storage tanks are equipped with hydrophobic bacteria retentive vent filters, the
filters should not be a source of contamination and the integrity of the filter tested before installation
and after use. Controls should be in place to prevent condensation formation on the filter (e.g. by
heating).
6.12 To minimize the risk of biofilm formation, sterilisation, disinfection or regeneration of water
systems should be carried out according to a predetermined schedule and as a remedial action
following out-of-limit or specification results. Disinfection of a water system with chemicals should
be followed by a validated rinsing/flushing procedure. Water should be tested after
disinfection/regeneration. Chemical testing results should be approved before the water system is
returned to use and microbiological/endotoxin results verified to be within specification and
approved before batches manufactured using water from the system are considered for
certification/release.
6.13 Regular ongoing chemical and microbial monitoring of water systems should be performed to
ensure that the water continues to meet compendial expectations. Alert levels should be based on the
initial qualification data and thereafter periodically reassessed on data obtained during subsequent
re-qualifications, routine monitoring, and investigations. Review of ongoing monitoring data should
be carried out to identify any adverse trend in system performance. Sampling programmes should
reflect the requirements of the CCS and should include all outlets and points of use, at a specified
interval, to ensure that representative water samples are obtained for analysis on a regular basis.
Sample plans should be based on the qualification data, should consider the potential worst case
sampling locations and should ensure that at least one representative sample is included every day of
the water that is used for manufacturing processes.
6.14 Alert level excursions should be documented and reviewed, and include an investigation to
determine whether the excursion is a single (isolated) event or if results are indicative of an adverse
trend or system deterioration. Each action limit excursion should be investigated to determine the
probable root causes and any potential impact on the quality of products and manufacturing
processes as a result of the use of the water.
6.15 WFI systems should include continuous monitoring systems such as Total Organic Carbon
(TOC) and conductivity, as these may give a better indication of overall system performance than
discrete sampling. Sensor locations should be based on risk.
Steam used as a direct sterilising agent
6.16 Feed water to a pure steam (clean steam) generator should be appropriately purified. Pure steam
generators should be designed, qualified and operated in a manner to ensure that the quality of steam
produced meets defined chemical and endotoxin levels.
6.17 Steam used as a direct sterilising agent should be of suitable quality and should not contain

18
additives at a level that could cause contamination of product or equipment. For a generator
supplying pure steam used for the direct sterilisation of materials or product-contact surfaces (e.g.
porous hard-good autoclave loads), steam condensate should meet the current monograph for WFI of
the relevant Pharmacopeia (microbial testing is not mandatory for steam condensate). A suitable
sampling schedule should be in place to ensure that representative pure steam is obtained for
analysis on a regular basis. Other aspects of the quality of pure steam used for sterilisation should be
assessed periodically against validated parameters. These parameters should include the following
(unless otherwise justified): non-condensable gases, dryness value (dryness fraction) and superheat.
Gases and vacuum systems
6.18 Gases that come in direct contact with the product/primary container surfaces should be of
appropriate chemical, particulate and microbial quality. All relevant parameters, including oil and
water content, should be specified, taking into account the use and type of the gas, the design of the
gas generation system and, where applicable, comply with the current monograph of the relevant
Pharmacopeia or the product quality requirement.
6.19 Gases used in aseptic processes should be filtered through a sterilising grade filter (with a
nominal pore size of a maximum of 0.22 µm) at the point of use. Where the filter is used on a batch
basis (e.g. for filtration of gas used for overlay of aseptically filled products) or as product vessel
vent filter, then the filter should be integrity tested and the results reviewed as part of the batch
certification/release process. Any transfer pipework or tubing that is located after the final sterilising
grade filter should be sterilised. When gases are used in the process, microbial monitoring of the gas
should be performed periodically at the point of use.
6.20 Where backflow from vacuum or pressure systems poses a potential risk to the product, there
should be mechanism(s) to prevent backflow when the vacuum or pressure system is shut off.
Heating and cooling and hydraulic systems
6.21 Major items of equipment associated with hydraulic, heating and cooling systems should, where
possible, be located outside the filling room. There should be appropriate controls to contain any
spillage and/or cross contamination associated with the system fluids.
6.22 Any leaks from these systems that would present a risk to the product should be detectable (e.g.
an indication system for leakage).
7 Personnel
7.1 The manufacturer should ensure that there are sufficient appropriate personnel, suitably qualified,
trained and experienced in the manufacture and testing of sterile products, and any of the specific
manufacturing technologies used in the site’s manufacturing operations, to ensure compliance with
GMP applicable to the manufacture and handling of sterile products.
7.2 Only the minimum number of personnel required should be present in cleanrooms. The
maximum number of operators in cleanrooms should be determined, documented and considered
during activities such as initial qualification and APS, so as not to compromise sterility assurance.
7.3 All personnel including those performing cleaning, maintenance, monitoring and those that
access cleanrooms should receive regular training, gowning qualification and assessment in
disciplines relevant to the correct manufacture of sterile products. This training should include the
basic elements of microbiology and hygiene, with a specific focus on cleanroom practices,
contamination control, aseptic techniques and the protection of sterile products (for those operators
entering the grade B cleanrooms and/or intervening into grade A) and the potential safety
implications to the patient if the product is not sterile. The level of training should be based on the
19
criticality of the function and area in which the personnel are working.
7.4 The personnel accessing grade A and B areas should be trained for aseptic gowning and aseptic
behaviours. Compliance with aseptic gowning procedures should be confirmed by assessment and
periodic reassessment at least annually, and should involve both visual and microbial assessment
(using monitoring locations such as gloved fingers, forearms, chest and hood (facemask / forehead).
See paragraph 9.30 for the expected limits). The unsupervised access to the grade A and grade B
areas where aseptic operations are or will be conducted should be restricted to appropriately
qualified personnel, who have passed the gowning assessment and have participated in a successful
APS.
7.5 Unqualified personnel should not enter grade B cleanrooms or grade A in operation. If needed in
exceptional cases, manufacturers should establish written procedures outlining the process by which
unqualified personnel are brought into the grade B and A areas. An authorized person from the
manufacturer should supervise the unqualified personnel during their activities and should assess the
impact of these activities on the cleanliness of the area. Access by these persons should be assessed
and recorded in accordance with the PQS.
7.6 There should be systems in place for the disqualification of personnel from working in or given
unsupervised entry into cleanrooms that is based on aspects including ongoing assessment and/or
identification of an adverse trend from the personnel monitoring programme and/or after being
implicated in a failed APS. Once disqualified, retraining and requalification should be completed
before permitting the operator to have any further involvement in aseptic practices. For operators
entering grade B cleanrooms or performing intervention into grade A, this requalification should
include consideration of participation in a successful APS.
7.7 High standards of personal hygiene and cleanliness are essential to prevent excessive shedding or
increased risk of introduction of microbial contamination. Personnel involved in the manufacture of
sterile products should be instructed to report any specific health conditions or ailments that may
cause the shedding of abnormal numbers or types of contaminants and therefore preclude cleanroom
access. Health conditions and actions to be taken with regard to personnel who could be introducing
an undue microbial hazard should be provided by the designated competent person and described in
procedures.
7.8 Personnel who have been engaged in the processing of human or animal tissue materials or of
cultures of micro-organisms, other than those used in the current manufacturing process, or any
activities that may have a negative impact to quality (e.g. microbial contamination), should not enter
clean areas unless clearly defined and effective decontamination and entry procedures have been
followed and documented.
7.9 Wristwatches, make-up, jewellery, other personal items such as mobile phones and any other
non-essential items should not be allowed in clean areas. Electronic devices used in cleanrooms, e.g.
mobile phones and tablets, that are supplied by the manufacturer solely for use in the cleanrooms,
may be acceptable if suitably designed to permit cleaning and disinfection commensurate with the
grade in which they are used. The use and disinfection of such equipment should be included in the
CCS.
7.10 Cleanroom gowning and hand washing should follow a written procedure designed to minimize
contamination of cleanroom clothing and/or the transfer of contaminants to the clean areas.
7.11 The clothing and its quality should be appropriate for the process and the grade of the
working area. It should be worn in such a way as to protect the product from contamination. When the
type of clothing chosen needs to provide the operator protection from the product, it should not
compromise the protection of the product from contamination. Garments should be visually checked
for cleanliness and integrity immediately prior to and after gowning. Gown integrity should also be
20
checked upon exit. For sterilised garments and eye coverings, particular attention should be taken to
ensure they have been subject to the sterilisation process, are within their specified hold time and that
the packaging is visually inspected to ensure it is integral before use. Reusable garments (including
eye coverings) should be replaced if damage is identified, or at a set frequency that is determined
during qualification studies. The qualification of garments should consider any necessary garment
testing requirements, including damage to garments that may not be identified by visual inspection
alone.
7.12 Clothing should be chosen to limit shedding due to operators’ movement.
7.13 A description of typical clothing required for each cleanliness grade is given below:
i. Grade B (including access / interventions into grade A): appropriate garments that are
dedicated for use under a sterilised suit should be worn before gowning (see paragraph 7.14).
Appropriately sterilised, non-powdered, rubber or plastic gloves should be worn while
donning the sterilised garments. Sterile headgear should enclose all hair (including facial hair)
and where separate from the rest of the gown, it should be tucked into the neck of the sterile
suit. A sterile facemask and sterile eye coverings (e.g. goggles) should be worn to cover and
enclose all facial skin and prevent the shedding of droplets and particles. Appropriate
sterilised footwear (e.g. over-boots) should be worn. Trouser legs should be tucked inside the
footwear. Garment sleeves should be tucked into a second pair of sterile gloves worn over the
pair worn while donning the gown. The protective clothing should minimize shedding of
fibres or particles and retain particles shed by the body. The particle shedding and the particle
retention efficiencies of the garments should be assessed during the garment qualification.
Garments should be packed and folded in such a way as to allow operators to don the gown
without contacting the outer surface of the garment and to prevent the garment from touching
the floor.
ii. Grade C: Hair, beards and moustaches should be covered. A single or two-piece trouser suit
gathered at the wrists and with high neck and appropriately disinfected shoes or overshoes
should be worn. They should minimize the shedding of fibres and particles.
iii. Grade D: Hair, beards and moustaches should be covered. A general protective suit and
appropriately disinfected shoes or overshoes should be worn. Appropriate measures should be
taken to avoid any ingress of contaminants from outside the clean area.
iv. Additional gowning including gloves and facemask may be required in grade C and D areas
when performing activities considered to be a contamination risk as defined by the CCS.
7.14 Cleanroom gowning should be performed in change rooms of an appropriate cleanliness grade
to ensure gown cleanliness is maintained. Outdoor clothing including socks (other than personal
underwear) should not be brought into changing rooms leading directly to grade B and C areas.
Single or two-piece facility trouser suits, covering the full length of the arms and the legs, and
facility socks covering the feet, should be worn before entry to change rooms for grades B and C.
Facility suits and socks should not present a risk of contamination to the gowning area or processes.
7.15 Every operator entering grade B or A areas should gown into clean, sterilised protective
garments (including eye coverings and masks) of an appropriate size at each entry. The maximum
period for which the sterilised gown may be worn before replacement during a shift should be
defined as part of the garment qualification.
7.16 Gloves should be regularly disinfected during operations. Garments and gloves should be
changed immediately if they become damaged and present any risk of product contamination.
7.17 Reusable clean area clothing should be cleaned in a laundry facility adequately segregated from
production operations, using a qualified process ensuring that the clothing is not damaged and/or
contaminated by fibres or particles during the repeated laundry process. Laundry facilities used
should not introduce risk of contamination or cross-contamination. Inappropriate handling and use of

21
clothing may damage fibres and increase the risk of shedding of particles. After washing and before
packing, garments should be visually inspected for damage and visual cleanliness. The garment
management processes should be evaluated and determined as part of the garment qualification
programme and should include a maximum number of laundry and sterilisation cycles.
7.18 Activities in clean areas that are not critical to the production processes should be kept to a
minimum, especially when aseptic operations are in progress. Movement of personnel should be
slow, controlled and methodical to avoid
excessive shedding of particles and organisms due to
over-vigorous activity. Operators performing aseptic operations should adhere to aseptic technique
at all times to prevent changes in air currents that may introduce air of lower quality into the critical
zone. Movement adjacent to the critical zone should be restricted and the obstruction of the path of
the unidirectional (first air) airflow should be avoided. A review of airflow visualisation studies
should be considered as part of the training programme.
8 Production and Specific Technologies
Terminally sterilised products
8.1 Preparation of components and materials should be performed in at least a grade D
cleanroom in order to limit the risk of microbial, endotoxin/pyrogen and particle contamination, so
that the product is suitable for sterilisation. Where the product is at a high or unusual risk of
microbial
contamination (e.g. the product actively supports microbial growth, the product must
be
held for long periods before filling or the product is not processed mostly in closed
vessels), then
preparation should be carried out in at least a grade C environment. Preparation of ointments,
creams, suspensions and emulsions should be carried out in at least a grade C environment before
terminal sterilisation. Specific guidance regarding terminally sterilised veterinary medicinal products
can be found within Annex 4 of the GMP guidelines.
8.2 Primary packaging containers and components should be cleaned using validated processes to
ensure that particle, endotoxin/pyrogen and bioburden contamination is appropriately controlled.
8.3 Filling of products for terminal sterilisation should be carried out in at least a grade C
environment.
8.4 Where the CCS identifies that the product is at an unusual risk of contamination from the
environment because, for example, the filling operation is slow, the containers are wide necked or are
necessarily exposed for more than a few seconds before closing, then the product should be filled in
grade A with at least a grade C background.
8.5 Processing of the bulk solution should include a filtration step with a microorganism retaining
filter, where possible, to reduce bioburden levels and particles prior to filling into the final product
containers and there should be a maximum permissible time between preparation and filling.
8.6 Examples of operations to be carried out in the various grades are given in Table 3.
Table 3: Examples of operations and grades for terminally sterilised preparation and
processing operations
Grade A
- Filling of products, when unusually at risk.
Grade C
- Preparation of solutions, when unusually at risk.
- Filling of products.
Grade D
- Preparation of solutions and components for subsequent filling.

22
Aseptic preparation and processing
8.7 The aseptic process should be clearly defined. The risks associated with the aseptic process, and
any associated requirements, should be identified, assessed and appropriately controlled. The site’s
CCS should clearly define the acceptance criteria for these controls, requirements for monitoring and
the review of their effectiveness. Methods and procedures to control these risks should be described
and implemented. Accepted residual risks should be formally documented.
8.8 Precautions to minimize microbial, endotoxin/pyrogenic and particle contamination should be
taken, as per the site’s CCS, during the preparation of the aseptic environment, during all processing
stages
(
including the stages before and after bulk product sterilisation), and until the product is sealed in
its final container. The presence of materials liable to generate particles and fibres should be minimized
in cleanrooms.
8.9 Where possible, the use of equipment such as RABS, isolators or other systems, should be
considered in order to reduce the need for critical interventions into grade A and to minimize the risk
of contamination. Robotics and automation of processes can also be considered to eliminate direct
human critical interventions (e.g. dry heat tunnel, automated lyophilizer loading, sterilisation in
place).
8.10 Examples of operations to be carried out in the various environmental grades are given in Table 4.
Table 4: Examples of operations and grades for aseptic preparation and processing operations
Grade A
- Aseptic assembly of filling equipment.
- Connections made under aseptic conditions (where sterilised product contact
surfaces are exposed) that are post the final sterilising grade filter. These
connections should be sterilised by steam-in-place whenever possible.
- Aseptic compounding and mixing.
- Replenishment of sterile bulk product, containers and closures.
- Removal and cooling of unprotected (e.g. with no packaging) items from
sterilisers.
- Staging and conveying of sterile primary packaging components in the aseptic
filling line while not wrapped.
- Aseptic filling, sealing of containers such as ampoules, vial closure, transfer
of open or partially stoppered vials.
- Loading of a lyophilizer.
Grade B
- Background support for grade A (when not in an isolator).
- Conveying or staging, while protected from the surrounding environment, of
equipment, components and ancillary items for introduction into grade A.
Grade C
- Preparation of solutions to be filtered including sampling and dispensing.
Grade D
- Cleaning of equipment.
- Handling of components, equipment and accessories after cleaning.
- Assembly under HEPA filtered airflow of cleaned components, equipment
and accessories prior to sterilisation.
- Assembly of closed and sterilised SUS using intrinsic sterile connection
devices.
23
8.11 For sterile products where the final formulation cannot be filtered, the following should be
considered:
i. All product and component contact equipment should be sterilised prior to use.
ii. All raw materials or intermediates should be sterilised and aseptically added.
iii. Bulk solutions or intermediates should be sterilised.
8.12 The unwrapping, assembly and preparation of sterilised equipment, components and ancillary
items with direct or indirect product contact should be treated as an aseptic process and performed in
grade A with a grade B background. The filling line set-up and filling of the sterile product should be
treated as an aseptic process and performed in grade A with a grade B background. Where an isolator
is used, the background should be in accordance with paragraph 4.20.
8.13 Preparation and filling of sterile products such as ointments, creams, suspensions and
emulsions should be
performed in grade A with a grade B background when the product and
components are exposed to the environment and
the product
is not subsequently filtered (via a
sterilising grade filter) or terminally sterilised. Where an isolator or RABS is used, the background
should be in accordance with paragraph 4.20.
8.14 Aseptic connections should be performed in grade A with a grade B background unless
subsequently sterilised in place or conducted with intrinsic sterile connection devices that minimize
any potential contamination from the immediate environment. Intrinsic sterile connection devices
should be designed to mitigate risk of contamination.
Where an isolator is used, the background should be in accordance with paragraph 4.20. Aseptic
connections should be appropriately assessed and their effectiveness verified. For requirements
regarding intrinsic sterile connection devices see paragraphs 8.129 and 8.130.
8.15 Aseptic manipulations (including non-intrinsic sterile connection devices) should be minimized
through the use of engineering design solutions such as preassembled and sterilised equipment.
Whenever feasible, product contact piping and equipment should be pre-assembled, and sterilised in
place.
8.16 There should be an authorized list of allowed and qualified interventions, both inherent and
corrective, that may occur during production (see paragraph 9.34). Interventions should be carefully
designed to ensure that the risk of contamination of the environment, process and product is
effectively minimized. The process of designing interventions should include the consideration of any
impact on air-flows and critical surfaces and products. Engineering solutions should be used
whenever possible to minimize incursion by operators during the intervention. Aseptic technique
should be observed at all times, including the appropriate use of sterile tools for manipulations. The
procedures listing the types of inherent and corrective interventions, and how to perform them, should
be first evaluated via risk management and APS and be kept up to date. Non-qualified interventions
should only be used in exceptional circumstances, with due consideration of the risks associated with
the intervention and with the authorisation of the quality unit. The details of the intervention
conducted should be subject to risk assessment, recorded and fully investigated under the
manufacturer's PQS. Any non-qualified interventions should be thoroughly assessed by the quality
department and considered during batch disposition.
8.17 Interventions and stoppages should be recorded in the batch record. Each line stoppage or
intervention should be sufficiently documented in batch records with the associated time, duration of
the event, and operators involved (ref to paragraph 9.34).
24
8.18 The duration of each aspect of aseptic preparation and processing should be minimized and
limited to a defined and validated maximum time, including:
i. The holding time between equipment, component, and container cleaning, drying and
sterilisation.
ii. The holding time for sterilised equipment, components, and containers before use and
during filling/assembly.
iii. The holding time for a decontaminated environment, such as the RABS or isolator before
use.
iv. The time between the start of the preparation of a product and its sterilisation or filtration
through a microorganism-retaining filter (if applicable), through to the end of the aseptic
filling process. There should be a maximum permissible time for each product that takes
into account its composition and the prescribed method of storage.
v. The holding time for sterilised product prior to filling.
vi. The aseptic processing time.
vii. The filling time.
8.19 Aseptic operations (including APS) should be observed on a regular basis by personnel with
specific expertise in aseptic processing to verify the correct performance of operations including
operator behaviour in the cleanroom and address inappropriate practices if detected.
Finishing of sterile products
8.20 Open primary packaging containers should be maintained under grade A conditions with the
appropriate background for the technology as described in paragraph 4.20. For partially stoppered
vials or prefilled syringes (see paragraph 8.126).
8.21 Final containers should be closed by appropriately validated methods.
8.22 Where final containers are closed by fusion, e.g. Blow-Fill-Seal (BFS), Form-Fill-Seal (FFS),
Small and Large Volume Parenteral (SVP & LVP) bags, glass or plastic ampoules, the critical
parameters and variables that affect seal integrity should be evaluated, determined, effectively
controlled and monitored during operations. Glass ampoules, BFS units and small volume
containers (≤100 ml) closed by fusion should be subject to 100% integrity testing using validated
methods. For large volume containers (>100 ml) closed by fusion, reduced sampling may be
acceptable where scientifically justified and based on data demonstrating the consistency of the
existing process, and a high level of process control. It should be noted that visual inspection is not
considered as an acceptable integrity test method.
8.23 Samples of products using systems other than fusion should be taken and checked for
integrity using validated methods. The frequency of testing should be based on the knowledge and
experience of the container and closure systems being used. A scientifically justified sampling
plan should be used. The sample size should be based on information such as supplier
management, packaging component specifications and process knowledge.
8.24 Containers sealed under vacuum should be tested for maintenance of vacuum after
an
appropriate pre-determined period prior to certification/release and during shelf life.
25
8.25 The container closure integrity validation should take into consideration any transportation or
shipping requirements that may negatively impact the integrity of the container (e.g. by
decompression or extreme temperatures).
8.26 Where the equipment used to crimp vial caps can generate large quantities of non-viable
particle, measures to prevent particle contamination such as locating the equipment at a physically
separate station equipped with adequate air extraction should be taken.
8.27 Vial capping of aseptically filled products can be undertaken as an aseptic process using
sterilised caps or as a clean
process outside the aseptic processing area. Where the latter
approach is adopted, vials should be
protected by grade A conditions up to the point of leaving
the aseptic processing area, and thereafter stoppered vials should be protected with a grade A air
supply until the cap has been crimped. The supporting background environment of grade A air
supply should meet at least grade D requirements. Where capping is a manual process, it should be
performed under grade A conditions either in an appropriately designed isolator or in grade A with a
grade B background.
8.28 Where capping of aseptically filled sterile product is conducted as a clean process with grade A
air supply protection, vials with missing or displaced stoppers should be rejected prior to capping.
Appropriately qualified, automated methods for stopper height detection should be in place.
8.29 Where
human intervention is required at the capping station, appropriate technological and
organizational measures should be used to prevent direct contact with the vials and to minimize
contamination. RABS and isolators may be beneficial in assuring the required
conditions
8.30 All filled containers of parenteral products should be inspected individually for extraneous
contamination or other defects. Defect classification and criticality should be determined during
qualification and based on risk and historical knowledge. Factors to consider include, but are not
limited to, the potential impact of the defect to the patient and the route of administration. Different
defect types should be categorized and batch performance analysed. Batches with unusual levels of
defects, when compared with routine defect numbers for the process (based on routine and trend
data), should be investigated. A defect library should be generated and maintained which captures all
known classes of defects. The defect library should be used for the training of production and quality
assurance personnel. Critical defects should not be identified during any subsequent sampling and
inspection of acceptable containers. Any critical defect identified subsequently should trigger an
investigation as it indicates a possible failure of the original inspection process.
8.31 When inspection is performed manually, it should be conducted under suitable and controlled
conditions of illumination and background. Inspection rates should be appropriately controlled and
qualified. Operators performing the inspection should undergo visual inspection qualification (whilst
wearing corrective lenses, if these are normally worn) at least annually. The qualification should be
undertaken using appropriate samples from the manufacturer's defect library sets and taking into
consideration worst case scenarios (e.g. inspection time, line speed where the product is transferred to
the operator by a conveyor system, container size or fatigue) and should include consideration of
eyesight checks. Operator distractions should be minimized and frequent breaks, of an appropriate
duration, should be taken from inspection.
8.32 Where automated methods of inspection are used, the process should be validated to detect
known defects (which may impact product quality or safety) and be equal to, or better than, manual
inspection methods. The performance of the equipment should be challenged using representative
defects prior to start up and at regular intervals throughout the batch.
8.33 Results of the inspection should be recorded and defect types and numbers trended. Reject levels
for the various defect types should also be trended based on statistical principles. Impact to product on
the market should be assessed as part of the investigation when adverse trends are observed.
26
Sterilisation
8.34 Where possible, finished product should be terminally sterilised, using a validated and controlled
sterilisation process, as this provides a greater assurance of sterility than a validated and controlled
sterile filtration process and/or aseptic processing. Where it is not possible for a product to undergo
terminal sterilisation, consideration should be given to using post-aseptic processing terminal heat
treatment, combined with aseptic process to give improved sterility assurance.
8.35 The selection, design and location of the equipment and cycle/programme used for sterilisation
should be based on scientific principles and data which demonstrate repeatability and reliability of the
sterilisation process. All parameters should be defined, and where critical, these should be controlled,
monitored and recorded.
8.36 All sterilisation processes should be validated. Validation studies should take into account the
product composition, storage conditions and maximum time between the start of the preparation of a
product or material to be sterilised and its sterilisation. Before any sterilisation process is adopted, its
suitability for the product and equipment, and its efficacy in consistently achieving the desired
sterilising conditions in all parts of each type of load to be processed should be validated notably by
physical measurements and where appropriate by Biological Indicators (BI). For effective
sterilisation, the whole of the product, and surfaces of equipment and components should be subject to
the required treatment and the process should be designed to ensure that this is achieved.
8.37 Particular attention should be given when the adopted product sterilisation method is not
described in the current edition of the Pharmacopoeia, or when it is used for a product which is not a
simple aqueous solution. Where possible, heat sterilisation is the method of choice.
8.38 Validated loading patterns should be established for all sterilisation processes and load patterns
should be subject to periodic revalidation. Maximum and minimum loads should also be considered as
part of the overall load validation strategy.
8.39 The validity of the sterilizing process should be reviewed and verified at scheduled intervals
based on risk. Heat sterilization cycles should be revalidated with a minimum frequency of at least
annually for load patterns that are considered worst case. Other load patterns should be validated at a
frequency justified in the CCS.
8.40 Routine operating parameters should be established and adhered to for all sterilisation
processes, e.g. physical parameters and loading patterns.
8.41 There should be mechanisms in place to detect a sterilisation cycle that does not conform to the
validated parameters. Any failed sterilisation or sterilisation that deviated from the validated process
(e.g. have longer or shorter phases such as heating cycles) should be investigated.
8.42 Suitable BIs placed at appropriate locations should be considered as an additional method to
support the validation of the sterilisation process. BIs should be stored and used according to the
manufacturer’s instructions. Where BIs are used to support validation and/or to monitor a
sterilisation process (e.g. with ethylene oxide), positive controls should be tested for each
sterilisation cycle. If BIs are used, strict precautions should be taken to avoid transferring microbial
contamination to the manufacturing or other testing processes. BI results in isolation should not be
used to override other critical parameters and process design elements.
8.43 The reliability of BIs is important. Suppliers should be qualified and transportation and storage
conditions should be controlled in order that BI quality is not compromised. Prior to use of a new
batch/lot of BIs, the population, purity and identity of the indicator organism of the batch/lot should
be verified. For other critical parameters, e.g. D-value, Z-value, the batch certificate provided by the
qualified supplier can normally be used.
27
8.44 There should be a clear means of differentiating products, equipment and components, which
have not been subjected to the sterilisation process from those which have. Equipment such as
baskets or trays used to carry products, other items of equipment and/or components should be
clearly labelled (or electronically tracked) with the product name and batch number and an
indication of whether or not it has been sterilised. Indicators such as autoclave tape, or irradiation
indicators may be used, where appropriate, to
indicate whether or not a batch (or sub-batch material,
component, equipment) has passed through a sterilisation process. However, these indicators show
only that the sterilisation process has occurred; they do not indicate product sterility or achievement
of the required sterility assurance level.
8.45 Sterilisation records should be available for each sterilisation run. Each cycle should have a
unique identifier. Their conformity should be reviewed and approved as part of the batch
certification/release procedure.
8.46 Where required, materials, equipment and components should be sterilised by validated methods
appropriate to the specific material. Suitable protection after sterilisation should be provided to
prevent recontamination. If sterilised items are not used immediately after sterilisation, these should
be stored using appropriately sealed packaging and a maximum hold time should be established.
Where justified, components that have been packaged with multiple sterile packaging layers need not
be stored in a cleanroom if the integrity and configuration of the sterile pack allows the items to be
readily disinfected during transfer by operators into grade A (e.g. by the use of multiple sterile
coverings that can be removed at each transfer from lower to higher grade). Where protection is
achieved by containment in sealed packaging, this packaging process should be undertaken prior to
sterilisation.
8.47 Where materials, equipment, components and ancillary items are sterilised in sealed packaging
and then transferred into grade A, this should be done using appropriate validated methods (for
example, airlocks or pass-through hatches) with accompanying disinfection of the exterior of the
sealed packaging. The use of rapid transfer port technology should also be considered. These methods
should be demonstrated to effectively control the potential risk of contamination of the grade A and
grade B areas and, likewise, the disinfection procedure should be demonstrated to be effective in
reducing any contamination on the packaging to acceptable levels for entry of the item into the grade
B and grade A areas.
8.48 Where materials, equipment, components and ancillary items are sterilised in sealed packaging
or containers, the packaging should be qualified for minimizing the risk of particulate, microbial,
endotoxin/pyrogen or chemical contamination, and for compatibility with the selected sterilisation
method. The packaging sealing process should be validated. The validation should consider the
integrity of the sterile protective barrier system, the maximum hold time before sterilisation and the
maximum shelf life assigned to the sterilised items. The integrity of the sterile protective barrier
system for each of the sterilised items should be checked prior to use.
8.49 For materials, equipment, components and ancillary items that are not a direct or indirect product
contact part and are necessary for aseptic processing but cannot be sterilised, an effective and
validated disinfection and transfer process should be in place. These items, once disinfected, should
be protected to prevent recontamination. These items, and others representing potential routes of
contamination, should be included in the environmental monitoring programme.
28
Sterilisation by heat
8.50 Each heat sterilisation cycle should be recorded either electronically or by hardcopy, using
equipment with suitable accuracy and
precision. The system should have safeguards and/or
redundancy in its control and monitoring instrumentation to detect a cycle not conforming to the
validated cycle parameter requirements and abort or fail this cycle (e.g. by the use of duplex/double
probes connected to independent control and monitoring systems).
8.51 The position of the temperature probes used for controlling and/or recording should be
determined during the validation and selected based on system design and in order to correctly
record and represent routine cycle conditions. Validation studies should be designed to demonstrate
the suitability of system control and recording probe locations, and should include the verification of
the function and location of these probes by the use of an independent monitoring probe located at
the same position during validation.
8.52 The whole of the load should reach the required temperature before measurement of the
sterilising time-period starts. For sterilisation cycles controlled by using a reference probe within the
load, specific consideration should be given to ensuring the load probe temperature is controlled
within defined temperature range prior to cycle commencement.
8.53 After completion of the high temperature phase of a heat sterilisation cycle, precautions should
be taken
against contamination of a sterilised load during cooling. Any cooling liquid or gas that
comes into contact with the product or sterilised material should be sterilised.
8.54 In those cases where parametric release has been authorized, a robust system should be applied
to the product lifecycle validation and the routine monitoring of the manufacturing process. This
system should be periodically reviewed. Further guidance regarding parametric release is provided in
Annex 17.
Moist heat sterilisation
8.55 Moist heat sterilisation can be achieved using steam, (direct or indirect contact), but also
includes other systems such as superheated water systems (cascade or immersion cycles) that could
be used for containers that may be damaged by other cycle designs (e.g. Blow-Fill-Seal containers,
plastic bags).
8.56 The items to be sterilised, other than products in sealed containers, should be dry, packaged in a
protective barrier system which allows removal of air and penetration of steam and prevents
recontamination after sterilisation. All loaded items should be dry upon removal from the steriliser.
Load dryness should be confirmed by visual inspection as a part of the sterilisation process
acceptance.
8.57 For porous cycles (hard goods), time, temperature and pressure should be used to monitor
the process and be recorded. Each sterilised item should be inspected for damage, packaging
material integrity and moisture on removal from the autoclave. Any item found not to be fit
for purpose should be removed from the manufacturing area and an investigation performed.
8.58 For autoclaves capable of performing prevacuum sterilisation cycles, the temperature
should be recorded at the chamber drain throughout the sterilisation period. Load probes may
also be used where appropriate but the controlling system should remain related to the load
validation. For steam in place systems, the temperature should be recorded at appropriate
condensate drain locations throughout the sterilisation period.
8.59 Validation of porous cycles should include a calculation of equilibration time, exposure time,
correlation of pressure and temperature and the minimum/maximum temperature range during
29
exposure. Validation of fluid cycles should include temperature, time and/or F
0
. Critical processing
parameters should be subject to defined limits (including appropriate tolerances) and be confirmed as
part of the sterilisation validation and routine cycle acceptance criteria.
8.60 Leak tests on the steriliser should be carried out periodically (normally weekly) when a vacuum
phase is part of the
cycle or the system is returned, post-sterilisation, to a pressure lower than the
environment surrounding the steriliser.
8.61 There should be adequate assurance of air removal prior to and during sterilisation when the
sterilisation process includes air purging (e.g. porous autoclave loads, lyophilizer chambers). For
autoclaves, this should include an air removal test cycle (normally performed on a daily basis) or the
use of an air detector system. Loads to be sterilised should be designed to support effective air
removal and be free draining to prevent the build-up of condensate.
8.62 Distortion and damage of non-rigid containers that are terminally sterilised, such as containers
produced by Blow-Fill-Seal or Form-Fill-Seal technologies, should be prevented by appropriate cycle
design and control (for instance setting correct pressure, heating and cooling rates and loading
patterns).
8.63 Where steam in place systems are used for sterilisation (e.g. for fixed pipework, vessels and
lyophilizer chambers), the system should be appropriately designed and validated to assure all parts
of the system are subjected to the required treatment. The system should be monitored for
temperature, pressure and time at appropriate locations during routine use to ensure all areas are
effectively and reproducibly sterilised. These locations should be demonstrated as being
representative of, and correlated with, the slowest to heat locations during initial and routine
validation. Once a system has been sterilised by steam in place, it should remain integral and where
operations require, maintained under positive pressure or otherwise equipped with a sterilising vent
filter prior to use.
8.64 In fluids load cycles where superheated water is used as the heat transfer medium, the heated
water should consistently reach all of the required contact points. Initial qualification studies should
include temperature mapping of the entire load. There should be routine checks on the equipment to
ensure that nozzles (where the water is introduced) are not blocked and drains remain free from
debris.
8.65 Validation of the sterilisation of fluids loads in a superheated water autoclave should include
temperature mapping of the entire load and heat penetration and reproducibility studies. All parts of
the load should heat up uniformly and achieve the desired temperature for the specified time.
Routine temperature monitoring probes should be correlated to the worst case positions identified
during the qualification process.
Dry heat sterilisation
8.66 Dry heat sterilisation utilizes high temperatures of air or gas to sterilise a product or article. Dry
heat sterilisation is of particular use in the thermal removal of difficult-to-eliminate thermally robust
contaminants such as endotoxin/pyrogen and is often used in the preparation of components for
aseptic filling. The combination of time and temperature to which product, components or equipment
are exposed should produce an adequate and reproducible level of lethality and/or endotoxin/pyrogen
inactivation/removal when operated routinely within the established limits. The process may be
operated in an oven or in a continuous tunnel process, e.g. for sterilisation and depyrogenation of
glass containers.
8.67 Dry heat sterilisation/depyrogenation tunnels should be configured to ensure that airflow protects
the integrity and performance of the grade A sterilising zone by maintaining appropriate pressure
differentials and airflow through the tunnel. Air pressure difference profiles should be assessed. The
30
impact of any airflow change should be assessed to ensure the heating profile is maintained. All air
supplied to the tunnel should pass through at least a HEPA filter and periodic tests (at least
biannually) should be performed to demonstrate air filter integrity. Any tunnel parts that come into
contact with sterilised components should be appropriately sterilised or disinfected. Critical process
parameters that should be considered during validation and/or routine processing should include, but
are not limited to:
i. Belt speed or dwell time within the sterilising zone.
ii. Temperature – minimum and maximum temperatures.
iii. Heat penetration of the material/article.
iv. Heat distribution/uniformity.
v. Airflows determined by air pressure difference profiles correlated with the heat distribution
and penetration studies.
8.68 When a thermal process is used as part of the depyrogenation process for any component or
product contact equipment/material, validation studies should be performed to demonstrate that the
process provides a suitable F
h
value and results in a minimum 3 log
10
reduction in endotoxin
concentration. When this is attained, there is no additional requirement to demonstrate sterilisation in
these cases.
8.69 Containers spiked with endotoxin should be used during validation and should be carefully
managed with a full reconciliation performed. Containers should be representative of the materials
normally processed (in respect to composition of the packaging materials, porosity, dimensions,
nominal volume). Endotoxin quantification and recovery efficiency should also be demonstrated.
8.70 Dry heat ovens are typically employed to sterilise or depyrogenate primary packaging
components, starting materials or active substances but may be used for other processes. They should
be maintained at a positive pressure relative to lower grade clean areas throughout the sterilisation and
post sterilisation hold process unless the integrity of the packaging is maintained. All air entering the
oven should pass through a HEPA filter. Critical process parameters that should be considered in
qualification and/or routine processing should include, but are not limited to:
i. Temperature.
ii. Exposure period/time.
iii. Chamber pressure (for maintenance of over pressure).
iv. Air speed.
v. Air quality within the oven.
vi. Heat penetration of material/article (slow to heat spots).
vii. Heat distribution/uniformity.
viii. Load pattern and configuration of articles to be sterilised/depyrogenated including minimum
and maximum loads.
31
Sterilisation by radiation
8.71 Sterilisation by radiation is used mainly for the sterilisation of heat sensitive materials and
products. Ultraviolet irradiation is not an acceptable method of sterilisation. Guidance regarding
ionising radiation sterilisation can be found within Annex 12.
8.72 Validation procedures should ensure that the effects of variation in density of the product and
packages are considered.
Sterilisation with ethylene oxide
8.73 This method should only be used when no other method is practicable. During process
validation, it should be shown that there is no damaging effect on the product and that the
conditions and time allowed for degassing result in the reduction of any residual ethylene oxide
(EO) gas and reaction
products to defined acceptable limits for the given product or material.
8.74 Direct contact between gas and microbial cells is essential, precautions should be taken to avoid
the presence of organisms likely to be enclosed in material such as crystals or dried protein. The
nature, porosity and quantity of packaging materials can significantly affect the process.
8.75 Before exposure to the gas, materials should be brought into equilibrium with the
humidity and temperature required by the process. Where steam is used to condition the load for
sterilisation, it should be of an appropriate quality. The time required for this should be
balanced
against the opposing need to minimize the time before sterilisation.
8.76 Each sterilisation cycle should be monitored with suitable BIs, using the
appropriate number of
test units distributed throughout the load at defined locations that have been shown to be worst case
locations during validation.
8.77 Critical process parameters that could be considered as part of the sterilisation process
validation and routine monitoring include, but are not limited to:
i. EO gas concentration.
ii. Pressure.
iii. Amount of EO gas used.
iv. Relative humidity.
v. Temperature.
vi. Exposure time.
8.78 After sterilisation, the load should be aerated to allow EO gas and/or its reaction products to
desorb from the packaged product to predetermined levels. Aeration can occur within a steriliser
chamber and/or in a separate aeration chamber or aeration room. The aeration phase should be
validated as part of the overall EO sterilisation process validation.
Filter sterilisation of products which cannot be sterilised in their final container
8.79 If the product cannot be sterilised in its final container, solutions or liquids should be sterilised
by filtration through a sterile sterilising grade filter (with a nominal pore size of a maximum of 0.22
µm that has been appropriately validated to obtain a sterile filtrate) and subsequently aseptically
filled into a previously sterilised container. The selection of the filter used should ensure that it is
32
compatible with the product and as described in the marketing authorization (see paragraph 8.135).
8.80 Suitable bioburden reduction prefilters and/or sterilising grade filters may be used at multiple
points during the manufacturing process to ensure a low and controlled bioburden of the liquid prior
to the final sterilising filter. Due to the potential additional risks of a sterile filtration process, as
compared with other sterilisation processes, an additional filtration through a sterile sterilising grade
filter, as close to the point of fill as possible, should be considered as part of an overall CCS.
8.81 The selection of components for the filtration system and their interconnection and arrangement
within the filtration system, including pre-filters, should be based on the critical quality attributes of
the product, justified and documented. The filtration system should minimize the generation of fibres
and particles, not cause or contribute to unacceptable levels of impurities, or possess characteristics
that otherwise alter the quality and efficacy of the product. Similarly, the filter characteristics should
be compatible with the fluid and not be adversely affected by the product to be filtered. Adsorption of
product components and extraction/leaching of filter components should be evaluated (see paragraph
8.135).
8.82 The filtration system should be designed to:
i. Allow operation within validated process parameters.
ii. Maintain the sterility of the filtrate.
iii. Minimize the number of aseptic connections required between the final sterilising grade
filter and the final filling of the product.
iv. Allow cleaning procedures to be conducted as necessary.
v. Allow sterilisation procedures, including sterilisation in place, to be conducted as necessary.
vi. Permit in-place integrity testing, of the 0.22 µm final sterilising grade filter, preferably as a
closed system, both prior to, and following filtration as necessary. In-place integrity testing
methods should be selected to avoid any adverse impact on the quality of the product.
8.83 Sterile filtration of liquids should be validated in accordance with relevant Pharmacopeia
requirements. Validation can be grouped by different strengths or variations of a product but should
be done under worst-case conditions. The rationale for grouping should be justified and documented.
8.84 During filter validation, wherever possible, the product to be filtered should be used for
bacterial retention testing of the sterilising grade filter. Where the product to be filtered is not
suitable for use in bacterial retention testing, a suitable surrogate product should be justified for use
in the test. The challenge organism used in the bacterial retention test should be justified.
8.85 Filtration parameters that should be considered and established during validation should
include, but are not limited to:
i. The wetting fluid used for filter integrity testing:
• It should be based on the filter manufacturer’s recommendation or the fluid to be
filtered. The appropriate integrity test value specification should be established.
• If the system is flushed or integrity tested in-situ with a fluid other than the product,
appropriate actions are taken to avoid any deleterious effect on product quality.
ii. Filtration process conditions including:
33
• Fluid pre-filtration holding time and effect on bioburden.
• Filter conditioning, with fluid if necessary.
• Maximum filtration time/total time filter is in contact with the fluid.
• Maximum operating pressure.
• Flow rate.
• Maximum filtration volume.
• Temperature.
• The time taken to filter a known volume of bulk solution and the
pressure difference
to be used across the filter.
8.86 Routine process controls should be implemented to ensure adherence to validated filtration
parameters. Results of critical process parameters should be included in the batch record, including
but not limited to the minimum time taken to filter a known volume of bulk solution and pressure
difference across the filter. Any significant difference from critical parameters during manufacturing
should be documented and investigated.
8.87 The integrity of the sterilised filter assembly should be verified by integrity testing before use
(pre-use post sterilisation integrity test or PUPSIT), to check for damage and loss of integrity caused
by the filter preparation prior to use. A sterilising grade filter that is used to sterilise a fluid should be
subject to a non-destructive integrity test post-use prior to removal of the filter from its housing. The
integrity test process should be validated and test results should correlate to the microbial retention
capability of the filter established during validation. Examples of tests that are used include bubble
point, diffusive flow, water intrusion or pressure hold test. It is recognized that PUPSIT may not
always be possible after sterilisation due to process constraints (e.g. the filtration of very small
volumes of solution). In these cases, an alternative approach may be taken providing that a thorough
risk assessment has been performed and compliance is achieved by the implementation of appropriate
controls to mitigate any risk of a non-integral filtration system. Points to consider in such a risk
assessment should include but are not limited to:
i. In depth knowledge and control of the filter sterilisation process to ensure that the potential
for damage to the filter is minimized.
ii. In depth knowledge and control of the supply chain to include:
• Contract sterilisation facilities.
• Defined transport mechanisms.
• Packaging of the sterilised filter, to prevent damage to the filter during transportation
and storage.
iii. In depth process knowledge such as:
• The specific product type, including particle burden and whether there exists any risk
of impact on filter integrity values, such as the potential to alter integrity-testing
values and therefore prevent the detection of a non-integral filter during a post-use
filter integrity test.
34
• Pre-filtration and processing steps, prior to the final sterilising grade filter, which
would remove particle burden and clarify the product prior to the sterile filtration.
8.88 The integrity of critical sterile gas and air vent filters (that are directly linked to the sterility of
the product) should be verified by testing after use, with the filter remaining in the filter assembly or
housing.
8.89 The integrity of non-critical air or gas vent filters should be confirmed and recorded at
appropriate intervals. Where gas filters are in place for extended periods, integrity testing should be
carried out at installation and prior to replacement. The maximum duration of use should be specified
and monitored based on risk (e.g. considering the maximum number of uses and heat treatment/
sterilisation cycles permitted as applicable).
8.90 For gas filtration, unintended moistening or wetting of the filter or filter equipment should be
avoided.
8.91 If the sterilising filtration process has been validated as a system consisting of multiple filters to
achieve the sterility for a given fluid, the filtration system is considered to be a single sterilising unit
and all filters within the system should satisfactorily pass integrity testing after use.
8.92 In a redundant filtration system (where a second redundant sterilising grade filter is present as a
backup but the sterilising process is validated as only requiring one filter), post-use integrity test of
the primary sterilising grade filter should be performed and if demonstrated to be integral, then a post-
use integrity test of the redundant (backup) filter is not necessary. However, in the event of a failure
of the post-use integrity test on the primary filter, post-use integrity test on the secondary (redundant)
filter should be performed, in conjunction with an investigation and risk assessment to determine the
reason for the primary filter test failure.
8.93 Bioburden samples should be taken from the bulk product and immediately prior to the final
sterile filtration. In case where a redundant filtration set-up is used, it should be taken prior to the first
filter. Systems for taking samples should be designed so as not to introduce contamination.
8.94 Liquid sterilising grade filters should be discarded after the processing of a single batch and the
same filter should not be used continuously for more than one working day unless such use has
been validated.
8.95 Where campaign manufacture of a product has been appropriately justified in the CCS and
validated, the filter user should:
i. Assess and document the risks associated with the duration of filter use for the sterile
filtration process for a given fluid.
ii. Conduct and document effective validation and qualification studies to demonstrate that the
duration of filter use for a given sterile filtration process and for a given fluid does not
compromise performance of the final sterilising grade filter or filtrate quality.
iii. Document the maximum validated duration of use for the filter and implement controls to
ensure that filters are not used beyond the validated maximum duration. Records of these
controls should be maintained.
iv. Implement controls to ensure that filters contaminated with fluid or cleaning agent residues,
or considered defective in any other way, are removed from use.
35
Form-Fill-Seal (FFS)
8.96 The conditions for FFS machines used for terminally sterilised products should comply with the
environmental requirements of paragraphs 8.3 and 8.4 of this Annex. The conditions for FFS
machines used in aseptic manufacture should comply with the environmental requirements of
paragraph 8.10 of this Annex.
8.97 Contamination of the packaging films used in the FFS process should be minimized by
appropriate controls during component fabrication, supply and handling. Due to the criticality of
packaging films, procedures should be implemented to ensure that the films supplied meet defined
specifications and are of the appropriate quality, including material thickness and strength, microbial
and particulate contamination, integrity and artwork, as relevant. The sampling frequency, the
bioburden and, where applicable, endotoxin/pyrogen levels of packaging films and associated
components should be defined and controlled within the PQS and considered in the CCS.
8.98 Particular attention should be given to understanding and assessing the operation of the
equipment, including set-up, filling, sealing and cutting processes, so that critical process parameters
are understood, validated, controlled and monitored appropriately.
8.99 Any product contact gases, e.g. those used to inflate the container or used as a product overlay,
should be appropriately filtered, as close to the point of use as possible. The quality of gases used and
the effectiveness of gas filtration systems should be verified periodically in accordance with
paragraphs 6.18 and 6.19.
8.100 The controls identified during qualification of FFS should be in alignment with the CCS.
Aspects to be considered include but are not limited to:
i. Determination of the boundaries of the critical zone.
ii. Environmental control and monitoring, both of the machine and the background in which it
is placed.
iii. Personnel gowning requirements.
iv. Integrity testing of the product filling lines and filtration systems (as relevant).
v. Duration of the batch or filling campaign.
vi. Control of packaging films, including any requirements for film decontamination or
sterilisation.
vii. Cleaning-in-place and sterilisation-in-place of equipment as necessary.
viii. Machine operation, settings and alarm management (as relevant).
8.101 Critical process parameters for FFS should be determined during equipment qualification and
should include, but are not limited to:
i. Settings for uniform package dimensions and cutting in accordance with validated
parameters.
ii. Setting, maintenance and monitoring of validated forming temperatures (including pre-
heating and cooling), forming times and pressures as relevant.
iii. Setting, maintenance and monitoring of validated sealing temperatures, sealing temperature
36
uniformity across the seal, sealing times and pressures as relevant.
iv. Environmental and product temperature.
v. Batch-specific testing of package seal strength and uniformity.
vi. Settings for correct filling volumes, speeds and uniformity.
vii. Settings for any additional printing (batch coding), embossing or debossing to ensure that
unit integrity is not compromised.
viii. Methods and parameters for integrity testing of filled containers (see paragraph 8.22).
8.102 Appropriate procedures for the verification, monitoring and recording of FFS critical process
parameters and equipment operation should be applied during production.
8.103 Operational procedures should describe how forming and sealing issues are detected and
rectified. Rejected units or sealing issues should be recorded and investigated.
8.104 Appropriate maintenance procedures should be established based on risk, and include
maintenance and inspection plans for tooling critical to the effectiveness of unit sealing. Any issues
identified that indicate a potential product quality concern should be documented and investigated.
Blow-Fill-Seal
8.105 Blow-Fill-Seal equipment used for the manufacture of products which are terminally sterilised
should be installed in at least a grade D environment. The conditions at the point of fill should comply
with the environmental requirements of paragraphs 8.3 and 8.4.
8.106 BFS used for aseptic processing:
i. For shuttle type equipment used for aseptic filling, the parison is open to the environment
and therefore the areas where parison extrusion, blow-moulding and sealing take place
should meet grade A conditions at the critical zones. The filling environment should be
designed and maintained to meet grade A conditions for viable and total particle limits both
at rest and when in operation.
ii. For rotary-type equipment used for aseptic filling, the parison is generally closed to the
environment once formed, the filling environment within the parison should be designed and
maintained to meet grade A conditions for viable and total particle limits both at rest and
when in operation.
iii. The equipment should be installed in at least a grade C environment, provided that grade
A/B clothing is used. The microbiological monitoring of operators wearing grade A/B
clothing in a grade C area, should be performed in accordance with risk management
principles, and the limits and monitoring frequencies applied with consideration of the
activities performed by these operators.
8.107 Due to the generation of particles from polymer extrusion and cutting during operation, and the
restrictive size of critical filling zones of BFS equipment, in operation monitoring of total particle for
BFS equipment is not expected. However, data should be available to demonstrate that the design of
the equipment ensures that critical zones of the filling process environment would meet grade A
conditions in operation.
37
8.108 Viable environmental monitoring of BFS processes should be risk-based, and designed in
accordance with section 9 of this Annex. In operation viable monitoring should be undertaken for the
full duration of critical processing, including equipment assembly. For rotary-type BFS equipment, it
is acknowledged that monitoring of the critical filling zone may not be possible.
8.109 The environmental control and monitoring programme should take into consideration the
moving parts and complex airflow paths generated by the BFS process and the effect of the high heat
outputs of the process, (e.g. through the use of airflow visualization studies and/or other equivalent
studies). Environmental monitoring programmes should also consider factors such as air-filter
configuration, air-filter integrity, cooling systems integrity (see paragraph 6.21), equipment design
and qualification.
8.110 Air or other gases that make contact with critical surfaces of the container during extrusion,
formation or sealing of the moulded container should undergo appropriate filtration. The quality of
gas used and the effectiveness of gas filtration systems should be verified periodically in accordance
with paragraphs 6.18 and 6.19.
8.111 Particulate and microbial contamination of the polymer granulate should be prevented by
appropriate design, control, and maintenance of the polymer granulate storage, sampling and
distribution systems.
8.112 The capability of the extrusion system to provide appropriate sterility assurance for the moulded
container should be understood and validated. The sampling frequency, the bioburden and, where
applicable, endotoxin/pyrogen levels of the raw polymer should be defined and controlled within the
PQS and considered in the CCS.
8.113 Interventions requiring cessation of filling and/or extrusion, moulding and sealing and, where
required, re-sterilisation of the filling machine should be clearly defined and described in the filling
procedure, and included in the APS as relevant (see paragraphs 9.34, 9.35 and 9.36).
8.114 The controls identified during qualification of BFS should be in alignment with the site’s CCS.
Aspects to be considered include but are not limited to:
i. Determination of the boundaries of the critical zone.
ii. Environmental control and monitoring, both of the machine and the background in which it
is placed.
iii. Personnel gowning requirements.
iv. Integrity testing of the product filling lines and filtration systems (as relevant).
v. Duration of the batch or filling campaign.
vi. Control of polymer granulate, including distribution systems and critical extrusion
temperatures.
vii. Cleaning-in-place and sterilisation-in-place of equipment as necessary.
viii. Machine operation, settings and alarm management (as relevant).
8.115 Critical process parameters for BFS should be determined during equipment qualification and
should include, but are not limited to:
38
i. Clean-in-place and sterilisation-in-place of product pipelines and filling needles (mandrels).
ii. Setting, maintenance and monitoring of extrusion parameters, including temperature, speed
and extruder throat settings for parison thickness.
iii. Setting, maintenance and monitoring of mould temperatures, including rate of cooling where
necessary for product stability.
iv. Preparation and sterilisation of ancillary components added to the moulded unit, e.g. bottle
caps.
v. Environmental control, cleaning, sterilisation and monitoring of the critical extrusion, transfer
and filling areas as relevant.
vi. Batch-specific testing of package wall-thickness at critical points of the container.
vii. Settings for correct filling volumes, speeds and uniformity.
viii. Settings for any additional printing (batch coding), embossing or debossing to ensure that unit
integrity and quality is not compromised.
ix. Methods and parameters for integrity testing of 100% of all filled containers (see paragraph
8.22).
x. Settings for cutters or punches used to remove waste plastic surrounding filled units (flash
removal).
8.116 Appropriate procedures for the verification, monitoring and recording of BFS critical process
parameters and equipment operation should be applied during production.
8.117 Operational procedures should describe how blowing, forming and sealing issues are detected
and rectified. Rejected units or sealing issues should be recorded and investigated.
8.118 Where the BFS process includes the addition of components to moulded containers (e.g.
addition of caps to LVP bottles), these components should be appropriately decontaminated and
added to the process using a clean, controlled process.
i. For aseptic processes, the addition of components should be performed under grade A
conditions, to ensure the sterility of critical surfaces, using pre-sterilised components.
ii. For terminally sterilised products, the validation of terminal sterilisation processes should
ensure the sterility of all critical product pathways between the component and moulded
container, including areas that are not wetted during sterilisation.
iii. Testing procedures should be established and validated to ensure the effective sealing of
components and moulded containers.
8.119 Appropriate maintenance procedures should be established based on risk, and include
maintenance and inspection plans for items critical to unit sealing, integrity and sterility.
8.120 The moulds used to form containers are considered critical equipment and any changes or
modification to moulds should result in an assessment of finished product container integrity, and
where the assessment indicates, should be supported by validation. Any issues identified that indicate
a potential product quality concern should be documented and investigated.
39
Lyophilization
8.121 Lyophilization is a critical process step and all activities that can affect the sterility of the
product or material need to be regarded as extensions of the aseptic processing of the sterilised
product. The lyophilization equipment and its processes should be designed to ensure that product or
material sterility is maintained during lyophilization by preventing microbial and particle
contamination between the filling of products for lyophilization, and completion of lyophilization
process. All control measures in place should be determined by the site’s CCS.
8.122 The sterilisation of the lyophilizer and associated equipment (e.g. trays, vial support rings)
should be validated and the holding time between the sterilisation cycle and use appropriately
challenged during APS (see paragraph 9.33). The lyophilizer should be sterilised regularly, based on
system design. Re-sterilisation should be performed following maintenance or cleaning. Sterilised
lyophilizers and associated equipment should be protected from contamination after sterilisation.
8.123 Lyophilizers and associated product transfer and loading/unloading areas should be designed to
minimize operator intervention as far as possible. The frequency of lyophilizer sterilisation should be
determined based on the design and risks related to system contamination during use. Lyophilizers
that are manually loaded or unloaded with no barrier technology separation should be sterilised before
each load. For lyophilizers loaded and unloaded by automated systems or protected by closed barrier
systems, the frequency of sterilisation should be justified and documented as part of the CCS.
8.124 The integrity of the lyophilizer should be maintained following sterilisation and during
lyophilization. The filter used to maintain lyophilizer integrity should be sterilised before each use of
the system and its integrity testing results should be part of the batch certification/release. The
frequency of vacuum/leak integrity testing of the chamber should be documented and the maximum
permitted leakage of air into the lyophilizer should be specified and checked at the start of every
cycle.
8.125 Lyophilization trays should be checked regularly to ensure that they are not misshapen or
damaged.
8.126 Points to consider for the design of loading (and unloading, where the lyophilised material is
still unsealed and exposed), include but are not limited to:
i. The loading pattern within the lyophilizer should be specified and documented.
ii. The transfer of partially closed containers to a lyophilizer should be undertaken under grade
A conditions at all times and handled in a manner designed to minimize direct operator
intervention. Technologies such as conveyor systems or portable transfer systems (e.g. clean
air transfer carts, portable unidirectional airflow workstations) should be used to ensure that
the cleanliness of the system used to transfer the partially closed containers is maintained.
Alternatively, where supported by validation, trays closed in grade A and not reopened whilst
in the grade B area may be used to protect partially stoppered vials (e.g. appropriately closed
boxes).
iii. Airflow patterns should not be adversely affected by transport devices and venting of the
loading zone.
iv. Unsealed containers (such as partially stoppered vials) should be maintained under grade A
conditions and should normally be separated from operators by physical barrier technology or
any other appropriate measures.
v. Where seating of the stoppers is not completed prior to opening the lyophilizer chamber,
product removed from the lyophilizer should remain under grade A conditions during
40
subsequent handling.
vi. Utensils used during loading and unloading of the lyophilizer (e.g. trays, bags, placing
devices, tweezers) should be sterile.
Closed systems
8.127 The use of closed systems can reduce the risk of microbial, particle and chemical contamination
from the adjacent environment. Closed systems should always be designed to reduce the need for
manual manipulations and the associated risks.
8.128 It is critical to ensure the sterility of all product contact surfaces of closed systems used for
aseptic processing. The design and selection of any closed system used for aseptic processing should
ensure maintenance of sterility. Connection of sterile equipment (e.g. tubing/pipework) to the
sterilised product pathway after the final sterilising grade filter should be designed to be connected
aseptically (e.g. by intrinsic sterile connection devices).
8.129 Appropriate measures should be in place to ensure the integrity of components used in aseptic
connections. The means by which this is achieved should be determined and captured in the CCS.
Appropriate system integrity tests should be considered when there is a risk of compromising product
sterility. Supplier assessment should include the collation of data in relation to potential failure modes
that may lead to a loss of system sterility.
8.130 The background environment in which closed systems are located should be based on their
design and the processes undertaken. For aseptic processing and where there are any risks that system
integrity may be compromised, the system should be located in grade A. If the system can be shown
to remain integral at every usage (e.g. via pressure testing and/or monitoring) then a lower classified
area may be used. Any transfer between classified areas should be thoroughly assessed (see paragraph
4.10). If the closed system is opened (e.g. for maintenance of a bulk manufacturing line) then this
should be performed in a classified area appropriate to the materials (e.g. grade C for terminal
sterilisation processes, or grade A for aseptic processing) or be subject to further cleaning and
disinfection (and sterilisation in case of aseptic processes).
Single use systems (SUS)
8.131 SUS are those technologies used in manufacture of sterile products which are used as an
alternative to reusable equipment. SUS can be individual components or made up of multiple
components such as bags, filters, tubing, connectors, valves, storage bottles and sensors. Single use
systems should be designed to reduce the need for manipulations and complexity of manual
interventions.
8.132 There are some specific risks associated with SUS which should be assessed as part of the CCS.
These risks include but are not limited to:
i. The interaction between the product and product contact surface (such as adsorption, or
leachables and extractables).
ii. The fragile nature of the system compared with fixed reusable systems.
iii. The increase in the number and complexity of manual operations (including inspection and
handling of the system) and connections made.
iv. The complexity of the assembly.
41
v. The performance of the pre- and post-use integrity testing for sterilising grade filters (see
paragraph 8.87).
vi. The risk of holes and leakage.
vii. The potential for compromising the system at the point of opening the outer packaging.
viii. The risk of particle contamination.
8.133 Sterilisation processes for SUS should be validated and shown to have no adverse impact on
system performance.
8.134 Assessment of suppliers of disposable systems including sterilisation is critical to the selection
and use of these systems. For sterile SUS, verification of sterility assurance should be performed as
part of the supplier qualification and evidence of sterilisation of each unit should be checked on
receipt.
8.135 The adsorption and reactivity of the product with product contact surfaces should be evaluated
under process conditions.
8.136 The extractable and leachable profiles of the SUS and any impact on the quality of the product
especially where the system is made from polymer-based materials should be evaluated. An
assessment should be carried out for each component to evaluate the applicability of the extractable
profile data. For components considered to be at high risk from leachables, including those that may
absorb processed materials or those with extended material contact times, an assessment of leachable
profile studies, including safety concerns, should be taken into consideration. If applying simulated
processing conditions, these should accurately reflect the actual processing conditions and be based
on a scientific rationale.
8.137 SUS should be designed to maintain integrity throughout processing under the intended
operational conditions. Attention to the structural integrity of the single use components is necessary
where these may be exposed to more extreme conditions (e.g. freezing and thawing processes) either
during routine processing or transportation. This should include verification that intrinsic sterile
connection devices (both heat sealed and mechanically sealed) remain integral under these conditions.
8.138 Acceptance criteria should be established and implemented for SUS corresponding to the risks
or criticality of the products and its processes. On receipt, each piece of SUS should be checked to
ensure that they have been manufactured, supplied and delivered in accordance with the approved
specification. A visual inspection of the outer packaging (e.g. appearance of exterior carton, product
pouches), label printing, and review of attached documents (e.g. certificate of conformance and proof
of sterilisation) should be carried out and documented prior to use.
8.139 Critical manual handling operations of SUS such as assembly and connections should be
subject to appropriate controls and verified during APS.

42
9 Environmental & process monitoring
General
9.1 The site’s environmental and process monitoring programme forms part of the overall CCS and is
used to monitor the controls designed to minimize the risk of microbial and particle contamination. It
should be noted that the reliability of each of the elements of the monitoring system (viable, non-
viable and APS) when taken in isolation is limited and should not be considered individually to be an
indicator of asepsis. When considered together, the results help confirm the reliability of the design,
validation and operation of the system that they are monitoring.
9.2 This programme is typically comprised of the following elements:
i. Environmental monitoring – total particle.
ii. Environmental and personnel monitoring – viable particle.
iii. Temperature, relative humidity and other specific characteristics.
iv. APS (aseptically manufactured product only).
9.3 The information from these systems should be used for routine batch certification/release and for
periodic assessment during process review or investigation. This applies for both terminal sterilisation
and aseptic processes, however, the criticality of the impact may differ depending upon the product
and process type.
Environmental and process monitoring
9.4 An environmental monitoring programme should be established and documented. The purpose of
the environmental monitoring programme, is to:
i. Provide assurance that cleanrooms and clean air equipment continue to provide an
environment of appropriate air cleanliness, in accordance with design and regulatory
requirements.
ii. Effectively detect excursions from environmental limits triggering investigation and
assessment of risk to product quality.
Risk assessments should be performed in order to establish this comprehensive environmental
monitoring programme, i.e. sampling locations, frequency of monitoring, monitoring methods and
incubation conditions (e.g. time, temperature(s), aerobic and/or anaerobic conditions).
These risk assessments should be conducted based on detailed knowledge of; the process inputs and
final product, the facility, equipment, the criticality of specific processes and steps, the operations
involved, routine monitoring data, monitoring data obtained during qualification and knowledge of
typical microbial flora isolated from the environment.
The risk assessment should include the determination of critical monitoring locations, those locations
where the presence of microorganisms during processing may have an impact upon product quality,
(e.g. grade A, aseptic processing areas and the grade B areas that directly interface with the grade A
area). Consideration of other information such as air visualisation studies should also be included.
These risk assessments should be reviewed regularly in order to confirm the effectiveness of the site’s
environmental monitoring programme. The monitoring programme should be considered in the
overall context of the trend analysis and the CCS for the site.
43
9.5 Routine monitoring of cleanrooms, clean air equipment and personnel should be performed in
operation throughout all critical stages of processing, including equipment set-up.
9.6 Other characteristics, such as temperature and relative humidity, should be controlled within
ranges that align with product/processing/personnel requirements and support maintenance of defined
cleanliness standards (e.g. grade A or B).
9.7 The monitoring of grade A should demonstrate the maintenance of aseptic processing conditions
during critical operations. Monitoring should be performed at locations posing the highest risk of
contamination to the sterile equipment surfaces, containers, closures and product. The selection of
monitoring locations and the orientation and positioning of sampling devices should be justified and
appropriate to obtain reliable data from the critical zones.
9.8 Sampling methods should not pose a risk of contamination to the manufacturing operations.
9.9 Appropriate alert levels and action limits should be set for the results of viable and total particle
monitoring. The maximum total particle action limits are described in Table 5 and the maximum
viable particle action limits are described in Table 6. However, more stringent action limits may be
applied based on data trending, the nature of the process or as determined within the CCS. Both viable
and total particle alert levels should be established based on results of cleanroom qualification tests
and periodically reviewed based on ongoing trend data.
9.10 Alert levels for grade A (total particle only) grade B, grade C and grade D should be set such that
adverse trends (e.g. a numbers of events or individual events that indicate a deterioration of
environmental control) are detected and addressed.
9.11 Monitoring procedures should define the approach to trending. Trends should include, but are not
limited to:
i. Increasing numbers of excursions from action limits or alert levels.
ii. Consecutive excursions from alert levels.
iii. Regular but isolated excursion from action limits that may have a common cause, (e.g.
single excursions that always follow planned preventative maintenance).
iv. Changes in microbial flora type and numbers and predominance of specific organisms.
Particular attention should be given to organisms recovered that may indicate a loss of
control, deterioration in cleanliness or organisms that may be difficult to control such as
spore-forming microorganisms and moulds.
9.12 The monitoring of grade C and D cleanrooms in operation should be performed based on data
collected during qualification and routine data to allow effective trend analysis. The requirements of
alert levels and action limits will depend on the nature of the operations carried out. Action limits
may be more stringent than those listed in Table 5 and Table 6.
9.13 If action limits are exceeded, operating procedures should prescribe a root cause investigation,
an assessment of the potential impact to product (including batches produced between the
monitoring and reporting) and requirements for corrective and preventive actions. If alert levels are
exceeded, operating procedures should prescribe assessment and follow-up, which should include
consideration of an investigation and/or corrective actions to avoid any further deterioration of the
environment.
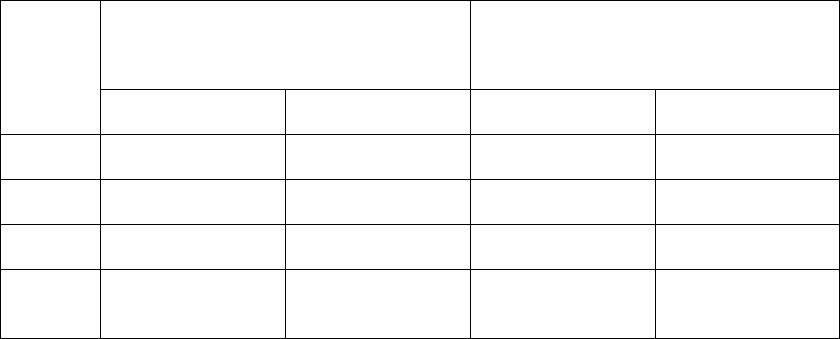
44
Environmental monitoring – total particle
9.14 A total particle monitoring program should be established to obtain data for assessing potential
contamination risks and to ensure the maintenance of the environment for sterile operations in a
qualified state.
9.15 The limits for environmental monitoring of airborne particle concentration for each graded
area are given in Table 5.
Table 5: Maximum permitted total particle concentration for monitoring.
Grade
Maximum limits for total particle
≥ 0.5 μm/m
3
Maximum limits for total particle
≥ 5 μm/m
3
at rest
in operation
at rest
in operation
A
3 520
3 520
29
29
B
3 520
352 000
29
2 930
C
352 000
3 520 000
2 930
29 300
D
3 520 000
Not
predetermined
(a)
29 300
Not
predetermined
(a)
(a)
For grade D, in operation limits are not predetermined. The manufacturer should establish in
operation limits based on a risk assessment and on routine data, where applicable.
Note 1: The particle limits given in the table for the “at rest” state should be achieved after a
short “clean up” period defined during qualification (guidance value of less than 20 minutes)
in an unmanned state, after the completion of operations (see paragraph 4.29).
Note 2: The occasional indication of macro particle counts, especially ≥ 5 µm, within grade A
may be considered to be false counts due to electronic noise, stray light, coincidence loss etc.
However, consecutive or regular counting of low levels may be indicative of a possible
contamination event and should be investigated. Such events may indicate early failure of the
room air supply filtration system, equipment failure, or may also be diagnostic of poor
practices during machine set-up and routine operation.
9.16 For grade A, particle monitoring should be undertaken for the full duration of critical
processing,
including equipment assembly.
9.17 The grade A area should be monitored continuously (for particles ≥0.5 and ≥5 µm) and with a
suitable sample flow rate (at least 28 litres (1ft
3
) per minute) so that all interventions, transient events
and any system deterioration is captured. The system should frequently correlate each individual
sample result with alert levels and action limits at such a frequency that any potential excursion can
be identified and responded to in a timely manner. Alarms should be triggered if alert levels are
exceeded. Procedures should define the actions to be taken in response to alarms including the
consideration of additional microbial monitoring.
9.18 It is recommended that a similar system be used for the grade B area although the sample
frequency may be decreased. The grade B area should be monitored at such a frequency and with
45
suitable sample size that the programme captures any increase in levels of contamination and system
deterioration. If alert levels are exceeded, alarms should be triggered.
9.19 The selection of the monitoring system should take into account any risk presented by the
materials used in the manufacturing operation (e.g. those involving live organisms, powdery
products or radiopharmaceuticals) that may give rise to biological, chemical or radiation hazards.
9.20 In the case where contaminants are present due to the processes involved and would potentially
damage the particle counter or present a hazard (e.g. live organisms, powdery products and radiation
hazards), the frequency and strategy employed should be such as to assure the environmental
classification both prior to and post exposure to the risk. An increase in viable particle monitoring
should be considered to ensure comprehensive monitoring of the process. Additionally, monitoring
should be performed during simulated operations. Such operations should be performed at appropriate
intervals. The approach should be defined in the CCS.
9.21 The size of monitoring samples taken using automated systems will usually be a
function of the
sampling rate of the system used. It is not necessary for the sample volume to
be the same as that
used for formal classification of cleanrooms and clean air equipment. Monitoring sample volumes
should be justified.
Environmental and personnel monitoring – viable particle
9.22 Where aseptic operations are performed, microbial monitoring should be frequent using a
combination of methods such as settle plates, volumetric air sampling, glove, gown and surface
sampling (e.g. swabs and contact plates). The method of sampling used should be justified within
the CCS and should be demonstrated not to have a detrimental impact on grade A and B airflow
patterns. Cleanroom and equipment surfaces should be monitored at the end of an operation.
9.23 Viable particle monitoring should also be performed within the cleanrooms when normal
manufacturing operations are not occurring (e.g. post disinfection, prior to start of manufacturing,
on completion of the batch and after a shutdown period), and in associated rooms that have not
been used, in order to detect potential incidents of contamination which may affect the controls
within the cleanrooms. In case of an incident, additional sample locations may be used as a
verification of the effectiveness of a corrective action (e.g. cleaning and disinfection).
9.24 Continuous viable air monitoring in grade A (e.g. air sampling or settle plates) should be
undertaken for the full duration of critical processing, including equipment (aseptic set-up)
assembly and critical processing. A similar approach should be considered for grade B cleanrooms
based on the risk of impact on the aseptic processing. The monitoring should be performed in such
a way that all interventions, transient events and any system deterioration would be captured and
any risk caused by interventions of the monitoring operations is avoided.
9.25 A risk assessment should evaluate the locations, type and frequency of personnel monitoring
based on the activities performed and the proximity to critical zones. Monitoring should include
sampling of personnel at periodic intervals during the process. Sampling of personnel should be
performed in such a way that it will not compromise the process. Particular consideration should be
given to monitoring personnel following involvement in critical interventions (at a minimum
gloves, but may require monitoring of areas of gown as applicable to the process) and on each exit
from the grade B cleanroom (gloves and gown). Where monitoring of gloves is performed after
critical interventions, the outer gloves should be replaced prior to continuation of activity. Where
monitoring of gowns is required after critical interventions, the gown should be replaced before
further activity in the cleanroom.
9.26 Microbial monitoring of personnel in the grade A and grade B areas should be performed.
Where operations are manual in nature (e.g. aseptic compounding or filling), the increased risk
46
should lead to enhanced emphasis placed on microbial monitoring of gowns and justified within the
CCS.
9.27 Where monitoring is routinely performed by manufacturing personnel, this should be subject to
regular oversight by the quality unit (refer also to paragraph 8.19).
9.28 The adoption of suitable alternative monitoring systems such as rapid methods should be
considered by manufacturers in order to expedite the detection of microbiological contamination
issues and to reduce the risk to product. These rapid and automated microbial monitoring methods
may be adopted after validation has demonstrated their equivalency or superiority to the established
methods.
9.29 Sampling methods and equipment used should be fully understood and procedures should be in
place for the correct operation and interpretation of results obtained. Supporting data for the recovery
efficiency of the sampling methods chosen should be available.
9.30 Action limits for viable particle contamination are shown in Table 6
Table 6: Maximum action limits for viable particle contamination
Grade
Air sample
CFU /m
3
Settle plates
(diam. 90 mm)
CFU /4 hours
(a)
Contact plates
(diam. 55mm),
CFU / plate
(b)
Glove print,
Including 5 fingers on
both hands
CFU / glove
A
No growth
(c)
B
10
5
5
5
C
100
50
25
-
D
200
100
50
-
(a)
- Settle plates should be exposed in grade A and B areas for the duration of operations
(including equipment set-up) and changed as required after a maximum of 4 hours (exposure
time should be based on validation including recovery studies and it should not have any
negative effect on the suitability of the media used).
- For grade C and D areas, exposure time (with a maximum of 4 hours) and frequency
should be based on QRM.
- Individual settle plates may be exposed for less than 4 hours.
(b)
Contact plate limits apply to equipment, room and gown surfaces within the grade A and
grade B areas. Routine gown monitoring is not normally required for grade C and D areas,
depending on their function.
(c)
It should be noted that for grade A, any growth should result in an investigation.
Note 1: It should be noted that the types of monitoring methods listed in the table above are
examples and other methods can be used provided they meet the intent of providing
information across the whole of the critical process where product may be contaminated (e.g.
aseptic line set-up, aseptic processing, filling and lyophilizer loading).
Note 2: Limits are applied using CFU throughout the document. If different or new
technologies are used that present results in a manner different from CFU, the manufacturer
should scientifically justify the limits applied and where possible correlate them to CFU.
47
9.31 Microorganisms detected in the grade A and grade B areas should be identified to species level
and the potential impact of such microorganisms on product quality (for each batch implicated) and
overall state of control should be evaluated. Consideration should also be given to the identification of
microorganisms detected in grade C and D areas (for example where action limits or alert levels are
exceeded) or following the isolation of organisms that may indicate a loss of control, deterioration in
cleanliness or that may be difficult to control such as spore-forming microorganisms and moulds and
at a sufficient frequency to maintain a current understanding of the typical flora of these areas.
Aseptic process simulation (APS) (also known as media fill)
9.32 Periodic verification of the effectiveness of the controls in place for aseptic processing should
include an APS using a sterile nutrient media and/or surrogate in place of the product. The APS
should not be considered as the primary means to validate the aseptic process or aspects of the aseptic
process. The effectiveness of the aseptic process should be determined through process design,
adherence to the pharmaceutical quality system and process controls, training, and evaluation of
monitoring data. Selection of an appropriate nutrient media and/or surrogate should be made based on
the ability of the media and/or surrogate to imitate physical product characteristics assessed to pose a
risk to product sterility during the aseptic process. Where processing stages may indirectly impact the
viability of any introduced microbial contamination, (e.g. aseptically produced semi-solids, powders,
solid materials, microspheres, liposomes and other formulations where product is cooled or heated or
lyophilized), alternative procedures that represent the operations as closely as possible should be
developed. Where surrogate materials, such as buffers, are used in parts of the APS, the surrogate
material should not inhibit the growth of any potential contamination.
9.33 The APS should imitate as closely as possible the routine aseptic manufacturing process and
include all the critical manufacturing steps, specifically:
i. The APS should assess all aseptic operations performed subsequent to the sterilisation and
decontamination cycles of materials utilised in the process to the point where the container
is sealed.
ii. For non-filterable formulations, any additional aseptic steps should be assessed.
iii. Where aseptic manufacturing is performed under an inert atmosphere, the inert gas should
be substituted with air in the process simulation unless anaerobic simulation is intended.
iv. Processes requiring the addition of sterile powders should use an acceptable surrogate
material in the same containers as those used in the process under evaluation.
v. Separate simulations of individual unit operations (e.g. processes involving drying,
blending, milling and subdivision of a sterile powder) should be avoided. Any use of
individual simulations should be supported by a documented justification and ensure that the
sum total of the individual simulations continues to fully cover the whole process.
vi. The process simulation procedure for lyophilized products should represent the entire
aseptic processing chain including filling, transport, loading, a representative duration of the
chamber dwell, unloading and sealing under specified, documented and justified conditions
representing worst case operating parameters.
vii. The lyophilization process simulation should mimic all aspects of the process, except those
that may affect the viability or recovery of contaminants. For instance, boiling-over or actual
freezing of the solution should be avoided. Factors to consider in determining APS design
include, where applicable:
• The use of air to break vacuum instead of nitrogen or other process gases.
48
• Replicating the maximum interval between sterilisation of the lyophilizer and its
use.
• Replicating the maximum period of time between filtration and lyophilization.
• Quantitative aspects of worst-case situations, e.g. loading the largest number of
trays, replicating the longest duration of loading where the chamber is open to the
environment.
9.34 The APS should take into account various aseptic manipulations and interventions known to
occur during normal production as well as worst-case situations, and take into account the following:
i. Inherent and corrective interventions representative of the routine process should be
performed in a manner and frequency similar to that during the routine aseptic process.
ii. The inclusion and frequency of interventions in the APS should be based on assessed risks
posed to product sterility.
9.35APS should not be used to justify practices that pose unnecessary contamination risks.
9.36 In developing the APS plan, consideration should be given to the following:
i. Identification of worst case conditions covering the relevant variables, such as container size
and line speed, and their impact on the process. The outcome of the assessment should
justify the variables selected.
ii. Determining the representative sizes of container/closure combinations to be used for
validation. Bracketing or matrix approach may be considered for validation of the same
container/closure configuration for different products where process equivalence is
scientifically justified.
iii. Maximum permitted holding times for sterile product and equipment exposed during the
aseptic process.
iv. The volume filled per container, which should be sufficient to ensure that the media contacts
all equipment and component surfaces that may directly contaminate the sterile product. The
volume used should provide sufficient headspace to support potential microbial growth and
ensure that turbidity can be detected during inspection.
v. The requirement for substitution of any inert gas used in the routine aseptic manufacturing
process by air unless anaerobic simulation is intended. In these situations, inclusion of
occasional anaerobic simulations as part of the overall validation strategy should be
considered (see paragraph 9.33 point iii).
vi. The selected nutrient media should be capable of growing a designated group of reference
microorganisms as described by the relevant pharmacopeia and suitably representative local
isolates.
vii. The method of detection of microbial contamination should be scientifically justified to
ensure that contamination is reliably detected.
viii. The process simulation should be of sufficient duration to challenge the process, the
operators that perform interventions, shift changes and the capability of the processing
49
environment to provide appropriate conditions for the manufacture of a sterile product.
ix. Where the manufacturer operates different or extended shifts, the APS should be designed to
capture factors specific to those shifts that are assessed to pose a risk to product sterility, for
example the maximum duration for which an operator may be present in the cleanroom.
x. Simulating normal aseptic manufacturing interruptions where the process is idle (e.g. shift
changeovers, recharging dispensing vessels, introduction of additional equipment).
xi. Ensuring that environmental monitoring is conducted as required for routine production, and
throughout the entire duration of the process simulation.
xii. Where campaign manufacturing occurs, such as in the use of Barrier Technologies or
manufacture of sterile active substances, consideration should be given to designing and
performing the process simulation so that it simulates the risks associated with both the
beginning and the end of the campaign and demonstrating that the campaign duration does
not pose any risk.
xiii. The performance of "end of production or campaign APS" may be used as additional
assurance or investigative purposes; however, their use should be justified in the CCS and
should not replace routine APS. If used, it should be demonstrated that any residual product
does not negatively impact the recovery of any potential microbial contamination.
9.37 For sterile active substances, batch size should be large enough to represent routine operation,
simulate intervention operation at the worst case, and cover all surfaces that may come into contact
with the sterile product. In addition, all the simulated materials (surrogates or growth medium) should
be subjected to microbial evaluation. The simulation materials should be sufficient to satisfy the
evaluation of the process being simulated and should not compromise the recovery of micro-
organisms.
9.38 APS should be performed as part of the initial validation, with at least three consecutive
satisfactory simulation tests that cover all working shifts that the aseptic process may occur in, and
after any significant modification to operational practices, facilities, services or equipment which are
assessed to have an impact on the sterility assurance of the product (e.g. modification to the HVAC
system, equipment, changes to process, number of shifts and numbers of personnel, major facility shut
down). Normally, APS (periodic revalidation) should be repeated twice a year (approximately every
six months) for each aseptic process, each filling line and each shift. Each operator should participate
in at least one successful APS annually. Consideration should be given to performing an APS after the
last batch prior to shut down, before long periods of inactivity or before decommissioning or
relocation of a line.
9.39 Where manual operation (e.g. aseptic compounding or filling) occurs, each type of container,
container closure and equipment train should be initially validated with each operator participating in
at least 3 consecutive successful APS and revalidated with one APS approximately every 6 months for
each operator. The APS batch size should mimic that used in the routine aseptic manufacturing
process.
9.40 The number of units processed (filled) for APS should be sufficient to effectively simulate all
activities that are representative of the aseptic manufacturing process. Justification for the number of
units to be filled should be clearly captured in the CCS. Typically, a minimum of 5000 to 10000 units
are filled. For small batches (e.g. those under 5000 units), the number of containers for APS should at
least equal the size of the production batch.
9.41 Filled APS units should be agitated, swirled or inverted before incubation to ensure contact of
the media with all interior surfaces in the container. All integral units from the APS should be
incubated and evaluated, including units with cosmetic defects or those which have gone through non-
50
destructive in-process control checks. If units are discarded during the process simulation and not
incubated, these should be comparable with units discarded during a routine fill, and only if
production SOPs clearly specify that units must be removed under the same circumstances (i.e. type
of intervention; line location; specific number of units removed). In no case should more units be
removed during a media fill intervention than would be cleared during a production run. Examples
may include those that must be discarded during routine production after the set-up process or
following a specific type of intervention. To fully understand the process and assess contamination
risks during aseptic setup or mandatory line clearances, these units would typically be incubated
separately, and would not necessarily be included in the acceptance criteria for the APS.
9.42 Where processes include materials that contact the product contact surfaces but are then
discarded (e.g. product flushes), the discarded material should be simulated with nutrient media and
be incubated as part of the APS, unless it can be clearly demonstrated that this waste process would
not impact the sterility of the product.
9.43 Filled APS units should be incubated in a clear container to ensure visual detection of microbial
growth. Where the product container is not clear (e.g. amber glass, opaque plastic), clear containers of
identical configuration may be substituted to aid in the detection of contamination. When a clear
container of identical configuration cannot be substituted, a suitable method for the detection of
microbial growth should be developed and validated. Microorganisms isolated from contaminated
units should be identified to the species level when practical, to assist in the determination of the
likely source of the contaminant.
9.44 Filled APS units should be incubated without unnecessary delay to achieve the best possible
recovery of potential contamination. The selection of the incubation conditions and duration should be
scientifically justified and validated to provide an appropriate level of sensitivity of detection of
microbial contamination.
9.45 On completion of incubation:
i. Filled APS units should be inspected by personnel who have been appropriately trained and
qualified for the detection of microbiological contamination. Inspection should be conducted
under conditions that facilitate the identification of any microbial contamination.
ii. Samples of the filled units should undergo positive control by inoculation with a suitable
range of reference organisms and suitably representative local isolates.
9.46 The target should be zero growth. Any contaminated unit should result in a failed APS and the
following actions should be taken:
i. An investigation to determine the most probable root cause(s).
ii. Determination and implementation of appropriate corrective measures.
iii. A sufficient number of successful, consecutive repeat APS (normally a minimum of 3) should
be conducted in order to demonstrate that the process has been returned to a state of control.
iv. A prompt review of all appropriate records relating to aseptic production since the last
successful APS.
a) The outcome of the review should include a risk assessment of potential sterile
breaches in batches manufactured since the last successful APS.
b) All other batches not released to the market should be included in the scope of the
investigation. Any decision regarding their release status should consider the
investigation outcome.

51
v. All products that have been manufactured on a line subsequent to a process simulation failure
should be quarantined until a successful resolution of the process simulation failure has
occurred.
vi. Where the root cause investigation indicates that the failure was related to operator activity,
actions to limit the operator’s activities, until retrained and requalified, should be taken.
vii. Production should resume only after completion of successful revalidation.
9.47 All APS runs should be fully documented and include a reconciliation of units processed (e.g.
units filled, incubated and not incubated). Justification for filled and non-incubated units should be
included in the documentation. All interventions performed during the APS should be recorded,
including the start and end time of each intervention and the involved person. All microbial
monitoring data as well as other testing data should be recorded in the APS batch record.
9.48 An APS run should be aborted only under circumstances in which written procedures require
commercial lots to be equally handled. An investigation should be documented in such cases.
9.49 An aseptic process should be subject to a repeat of the initial validation when:
i. The specific aseptic process has not been in operation for an extended period of time.
ii. There is a change to the process, equipment, procedures or environment that has the
potential to affect the aseptic process or an addition of new product containers or container-
closure combinations.
10 Quality Control (QC)
10.1 There should be personnel available with appropriate training and experience in microbiology,
sterility assurance and knowledge of the processes to support the design of the manufacturing
activities, environmental monitoring regime and any investigation assessing the impact of
microbiologically linked events to the safety of the sterile product.
10.2 Specifications for raw materials, components and products should include requirements for
microbial, particulate and endotoxin/pyrogen limits when the need for this has been indicated by
monitoring and/or by the CCS.
10.3 The bioburden assay should be performed on each batch for both aseptically filled product and
terminally sterilised products and the results considered as part of the final batch review. There should
be defined limits for bioburden immediately before the final sterilising grade filter or the terminal
sterilisation process, which are related to the efficiency of the method to be used. Samples should be
taken to be representative of the worst-case scenario (e.g. at the end of hold time). Where overkill
sterilisation parameters are set for terminally sterilised products, bioburden should be monitored at
suitable scheduled intervals.
10.4 For products authorised for parametric release, a supporting pre-sterilisation bioburden
monitoring programme for the filled product prior to initiating the sterilisation cycle should be
developed and the bioburden assay should be performed for each batch. The sampling locations of
filled units before sterilisation should be based on a worst case scenario and be representative of the
batch. Any organisms found during bioburden testing should be identified and their impact on the
effectiveness of the sterilising process determined. Where appropriate, the level of
endotoxin/pyrogen should be monitored.
52
10.5 The sterility test applied to the finished product should only be regarded as the last in a series of
critical control measures by which sterility is assured. It cannot be used to assure sterility of a
product that does not meet its design, procedural or validation parameters. The test should be
validated for the product concerned.
10.6 The sterility test should be performed under aseptic conditions. Samples taken for sterility
testing should be representative of the whole of the batch but should in particular include samples
taken from parts of the batch considered to be most at risk of contamination, for example:
i. For products which have been filled aseptically, samples should include containers filled at
the beginning and end of the batch. Additional samples, e.g. taken after critical interventions
should be considered based on risk.
ii. For products which have been heat sterilised in their final containers, samples taken
should
be
representative of the worst case locations (e.g. the potentially coolest or slowest to
heat part of each load).
iii. For products which have been lyophilized, samples taken from different lyophilization
loads.
Note: Where the manufacturing process results in sub-batches (e.g. for terminally sterilised products)
then sterility samples from each sub-batch should be taken and a sterility test for each sub-batch
performed. Consideration should also be given to performing separate testing for other finished
product tests.
10.7 For some products it may not be possible to obtain a sterility test result prior to release because
the shelf life of the product is too short to allow completion of a sterility test. In these cases, the
additional considerations of design of the process and additional monitoring and/or alternative test
methods required to mitigate the identified risks should be assessed and documented.
10.8 Any process (e.g. Vaporized Hydrogen Peroxide, Ultra Violet) used to decontaminate the
external surfaces of sterility samples prior to testing should not negatively impact the sensitivity of the
test method or the reliability of the sample.
10.9 Media used for product testing should be quality control tested according to the related
Pharmacopeia before use. Media used for environmental monitoring and APS should be tested for
growth promotion before use, using a scientifically justified and designated group of reference
microorganisms and including suitably representative local isolates. Media quality control testing
should normally be performed by the end user. Any reliance on outsourced testing or supplier testing
of media should be justified and transportation and shipping conditions should be thoroughly
considered in this case.
10.10 Environmental monitoring data and trend data generated for classified areas should be reviewed
as part of product batch certification/release. A written procedure should be available that describes
the actions to be taken when data from environmental monitoring are found out of trend or exceeding
the established limits. For products with short shelf life, the environmental data for the time of
manufacture may not be available; in these cases, the compliance should include a review of the most
recent available data. Manufacturers of these products should consider the use of rapid/alternative
methods.
10.11 Where rapid and automated microbial methods are used for general manufacturing purposes,
these methods should be validated for the product(s) or processes concerned.

53
Glossary
Airlock – An enclosed space with interlocked doors, constructed to maintain air pressure control
between adjoining rooms (generally with different air cleanliness standards). The intent of an airlock
is to preclude ingress of particle matter and microorganism contamination from a lesser controlled
area.
Action limit – An established relevant measure (e.g. microbial, or airborne particle limits) that, when
exceeded, should trigger appropriate investigation and corrective action based on the investigation.
Alert level – An established relevant measure (e.g. microbial, or airborne particle levels) giving early
warning of potential drift from normal operating conditions and validated state, which does not
necessarily give grounds for corrective action but triggers appropriate scrutiny and follow-up to
address the potential problem. Alert levels are established based on routine and qualification trend
data and are periodically reviewed. The alert level can be based on a number of parameters including
adverse trends, individual excursions above a set limit and repeat events.
Aseptic preparation/processing – The handling of sterile product, containers and/or devices in a
controlled environment in which the air supply, materials and personnel are regulated to prevent
microbial, endotoxin/pyrogen and particle contamination.
Aseptic Process Simulation (APS) – A simulation of the entire aseptic manufacturing process in order
to verify the capability of the process to assure product sterility. Includes all aseptic operations
associated with routine manufacturing, e.g. equipment assembly, formulation, filling, lyophilization
and sealing processes as necessary.
Asepsis – A state of control attained by using an aseptic work area and performing activities in a
manner that precludes microbial contamination of the exposed sterile product.
Bacterial retention testing – This test is performed to validate that a filter can remove bacteria from a
gas or liquid. The test is usually performed using a standard organism, such as Brevundimonas
diminuta at a minimum concentration of 10
7
Colony Forming Units/cm
2
.
Barrier – A physical partition that affords aseptic processing area (usually grade A) protection by
separating it from the background environment. Such systems frequently use in part or totally the
Barrier Technologies known as RABS or isolators.
Bioburden – The total number of microorganisms associated with a specific item such as personnel,
manufacturing environments (air and surfaces), equipment, product packaging, raw materials
(including water), in-process materials, or finished products.
Bio-decontamination - A process that eliminates viable bioburden via use of sporicidal chemical
agents.
Biological Indicators (BI) – A population of microorganisms inoculated onto a suitable medium (e.g.
solution, container or closure) and placed within a steriliser or load or room locations to determine the
sterilisation or disinfection cycle efficacy of a physical or chemical process. The challenge
microorganism is selected and validated based upon its resistance to the given process. Incoming lot
D-value, microbiological count and purity define the quality of the BI.
Blow-Fill-Seal (BFS) – A technology in which containers are formed from a thermoplastic granulate,
filled with product, and then sealed in a continuous, integrated, automatic operation. The two most
common types of BFS machines are the Shuttle type (with Parison cut) and the Rotary type (Closed
Parison).

54
Campaign manufacture – A manufacture of a series of batches of the same product in sequence in a
given period of time with strict adherence to established and validated control measures.
Classified area – An area that contains a number of cleanrooms (see cleanroom definition).
Cleaning – A process for removing contamination e.g. product residues or disinfectant residues.
Clean area – An area with defined particle and microbiological cleanliness standards usually
containing a number of joined cleanrooms.
Cleanroom – A room designed, maintained, and controlled to prevent particle and microbial
contamination of drug products. Such a room is assigned and reproducibly meets an appropriate air
cleanliness level.
Cleanroom classification – A method of assessing the level of air cleanliness against a specification
for a cleanroom or clean air equipment by measuring the total particle concentration.
Cleanroom qualification – A method of assessing the level of compliance of a classified cleanroom or
clean air equipment with its intended use.
Closed system – A system in which the product is not exposed to the surrounding environment. For
example, this can be achieved by the use of bulk product holders (such as tanks or bags) that are
connected to each other by pipes or tubes as a system, and where used for sterile products, the full
system is sterilised after the connections are made. Examples of these can be (but are not limited to)
large scale reusable systems, such as those seen in active substance manufacturing, or disposable bag
and manifold systems, such as those seen in the manufacture of biological products. Closed systems
are not opened until the conclusion of an operation. The use of the term “closed systems” in this
Annex does not refer to systems such as RABS or isolator systems.
Colony Forming Unit (CFU) – A microbiological term that describes a single detectable colony that
originates from one or more microorganisms. Colony forming units are typically expressed as CFU
per ml for liquid samples, CFU per m
3
for air sample and CFU per sample for samples captured on
solid medium such as settle or contact plates.
Contamination – The undesired introduction of impurities of a microbiological nature (quantity and
type of microorganisms, pyrogen), or of foreign particle matter, into or onto a raw material,
intermediate, active substance or drug product during production, sampling, packaging or
repackaging, storage or transport with the potential to adversely impact product quality.
Contamination Control Strategy (CCS) – A planned set of controls for microorganisms,
endotoxin/pyrogen and particles, derived from current product and process understanding that assures
process performance and product quality. The controls can include parameters and attributes related to
active substance, excipient and drug product materials and components, facility and equipment
operating conditions, in-process controls, finished product specifications, and the associated methods
and frequency of monitoring and control.
Corrective intervention – An intervention that is performed to correct or adjust an aseptic process
during its execution. These may not occur at a set frequency in the routine aseptic process. Examples
include such as clearing component jams, stopping leaks, adjusting sensors, and replacing equipment
components.
Critical surfaces – Surfaces that may come directly into contact with, or directly affect, a sterile
product or its containers or closures. Critical surfaces are rendered sterile prior to the start of the
manufacturing operation, and sterility is maintained throughout processing.

55
Critical zone – A location within the aseptic processing area in which product and critical surfaces are
exposed to the environment.
Critical intervention – An intervention (corrective or inherent) into the critical zone.
D-value – The value of a parameter of sterilisation (duration or absorbed dose) required to reduce the
number of viable organisms to 10 per cent of the original number.
Dead leg – Length of non-circulating pipe (where fluid may remain static) that is greater than 3
internal pipe diameters.
Decommission – When a process, equipment or cleanroom are closed and they will not be used again.
Decontamination – The overall process of removal or reduction of any contaminants (chemical, waste,
residue or microorganisms) from an area, object, or person. The method of decontamination used (e.g.
cleaning, disinfection, sterilisation) should be chosen and validated to achieve a level of cleanliness
appropriate to the intended use of the item decontaminated. See also Bio-decontamination.
Depyrogenation – A process designed to remove or inactivate pyrogenic material (e.g. endotoxin) to a
specified minimum quantity.
Disinfection – The process by which the reduction of the number of microorganisms is achieved by
the irreversible action of a product on their structure or metabolism, to a level deemed to be
appropriate for a defined purpose.
Endotoxin – A pyrogenic product (i.e. lipopolysaccharide) present in the Gram negative bacterial cell
wall. Endotoxin can lead to reactions in patients receiving injections ranging from fever to death.
Equilibration time – Period which elapses between the attainment of the sterilisation temperature at
the reference measurement point and the attainment of the sterilisation temperature at all points within
the load.
Extractables - Chemical entities that migrate from the surface of the process equipment, exposed to an
appropriate solvent at extreme conditions, into the product or material being processed.
First Air – Refers to filtered air that has not been interrupted prior to contacting exposed product and
product contact surfaces with the potential to add contamination to the air prior to reaching the critical
zone.
Filter Integrity test - A test to confirm that a filter (product, gas or HVAC filter) retain their retentive
properties and have not been damaged during handling, installation or processing.
Form-Fill-Seal (FFS) –An automated filling process, typically used for terminally sterilised products,
which constructs the primary container out of a continuous flat roll of packaging film while
simultaneously filling the formed container with product and sealing the filled containers in a
continuous process. FFS processes may utilize a single web system (where a single flat roll of film is
wrapped around itself to form a cavity), or a dual web system (where two flat rolls of film are brought
together to form a cavity), often with the aid of vacuum moulds or pressurised gases. The formed
cavity is filled, sealed and cut into sections. Films typically consist of a polymeric material, polymeric
coated foil or other suitable material.
Gowning qualification – A programme that establishes, both initially and on a periodic basis, the
capability of an individual to don the complete gown.

56
Grade A air supply – Air which is passed through a filter qualified as capable of producing grade A
total particle quality air, but where there is no requirement to perform continuous total particle
monitoring or meet grade A viable monitoring limits. Specifically used for the protection of fully
stoppered vials where the cap has not yet been crimped.
HEPA filter – High efficiency particulate air filter specified in accordance with a relevant
international standard.
Inherent interventions – An intervention that is an integral part of the aseptic process and is required
for either set-up, routine operation and/or monitoring (e.g. aseptic assembly, container replenishment,
environmental sampling). Inherent interventions are required by procedure or work instruction for the
execution of the aseptic process.
Intrinsic sterile connection device – A device that reduces the risk of contamination during the
connection process; these can be mechanical or fusion sealing.
Isokinetic sampling head – A sampling head designed to disturb the air as little as possible so that the
same particles go into the nozzle as would have passed the area if the nozzle had not been there (i.e.
the sampling condition in which the mean velocity of the air entering the sample probe inlet is nearly
the same (± 20 percent) as the mean velocity of the airflow at that location).
Isolator – An enclosure capable of being subject to reproducible interior bio-decontamination, with an
internal work zone meeting grade A conditions that provides uncompromised, continuous isolation of
its interior from the external environment (e.g. surrounding cleanroom air and personnel). There are
two major types of isolators:
i. Closed isolator systems exclude external contamination of the isolator’s interior by
accomplishing material transfer via aseptic connection to auxiliary equipment, rather
than use of openings to the surrounding environment. Closed systems remain sealed
throughout operations.
ii. Open isolator systems are designed to allow for the continuous or semi-continuous
ingress and/or egress of materials during operations through one or more openings.
Openings are engineered (e.g. using continuous overpressure) to exclude the entry of
external contaminant into the isolator.
Leachables – Chemical entities that migrate into products from the product contact surface of the
process equipment or containers under normal condition of use and/or storage.
Local isolates – Suitably representative microorganisms of the site that are frequently recovered
through environmental monitoring within the classified zone/areas especially grade A and B areas,
personnel monitoring or positive sterility test results.
Lyophilization – A physical-chemical drying process designed to remove solvents, by way of
sublimation, from both aqueous and non-aqueous systems, primarily to achieve product or material
stability. Lyophilization is synonymous to the term freeze-drying.
Manual aseptic processing– An aseptic process where the operator manually compounds, fills, places
and /or seals an open container with sterile product.
Operator - Any individual participating in the processing operation, including line set-up, filling,
maintenance, or other personnel associated with manufacturing activities.
Overkill sterilisation – A process that is sufficient to provide at least a 12 log
10
reduction of
microorganisms having a minimum D-value of 1 minute.

57
Parison – The "tube" of polymer extruded by the BFS machine from which containers are formed.
Pass-through hatch – Synonymous with airlock (see airlock definition) but typically smaller in size.
Patient – Human or animal including participants in a clinical trial.
Post-aseptic processing terminal heat treatment– A terminal moist heat process employed after aseptic
processing which has been demonstrated to provide a sterility assurance level (SAL) ≤10
-6
but where
the requirements of steam sterilisation (for example, F
0
≥8 min) are not fulfilled. This may also be
beneficial in the destruction of viruses that may not be removed through filtration.
Pyrogen – A substance that induces a febrile reaction in patients receiving injections;
Rapid Transfer System/Port (RTP) – A System used for the transfer of items into RABS or isolators
that minimizes the risk to the critical zone. An example would be a rapid transfer container with an
alpha/beta port.
Raw material – Any ingredient intended for use in the manufacture of a sterile product, including
those that may not appear in the final drug product.
Restricted Access Barrier System (RABS) – System that provides an enclosed, but not fully sealed,
environment meeting defined air quality conditions (for aseptic processing grade A), and using a
rigid-wall enclosure and integrated gloves to separate its interior from the surrounding cleanroom
environment. The inner surfaces of the RABS are disinfected and decontaminated with a sporicidal
agent. Operators use gloves, half suits, RTPs and other integrated transfer ports to perform
manipulations or convey materials to the interior of the RABS. Depending on the design, doors are
rarely opened, and only under strictly pre-defined conditions.
Single Use Systems (SUS) – Systems in which product contact components are used only once to
replace reusable equipment such as stainless steel transfer lines or bulk containers. SUS covered in
this document are those that are used in manufacturing processes of sterile products and are typically
made up of disposable components such as bags, filters, tubing, connectors, storage bottles and
sensors.
Sporicidal agent – An agent that destroys bacterial and fungal spores when used in sufficient
concentration for specified contact time. It is expected to kill all vegetative microorganisms.
Sterile Product – For purpose of this guidance, sterile product refers to one or more of the sterilised
elements exposed to aseptic conditions and ultimately making up the sterile active substance or
finished sterile product. These elements include the containers, closures, and components of the
finished drug product. Or, a product that is rendered sterile by a terminal sterilisation process.
Sterilising grade filter – A filter that, when appropriately validated, will remove a defined microbial
challenge from a fluid or gas producing a sterile effluent. Usually such filters have a pore size equal or
less than 0.22 µm.
Terminal Sterilisation – The application of a lethal sterilising agent or conditions to a product in its
final container to achieve a predetermined sterility assurance level (SAL) of 10⁻⁶ or better (e.g. the
theoretical probability of there being a single viable microorganism present on or in a sterilised unit is
equal to or less than 1 x 10
-6
(one in a million)).
Turbulent airflow – Air that is not unidirectional. Turbulent air in cleanrooms should flush the
cleanroom via mixed flow dilution and ensure maintenance of acceptable air quality.

58
Unidirectional airflow – An airflow moving in a single direction, in a robust and uniform manner, and
at sufficient speed, to reproducibly sweep particles away from the critical processing or testing area.
Unidirectional Airflow (UDAF) unit – A cabinet supplied with filtered unidirectional airflow
(previously referred to as a Laminar Airflow Unit or LAF).
Worst case – A set of conditions encompassing processing limits and circumstances, including those
within standard operating procedures, that pose the greatest chance of process or product failure
(when compared with ideal conditions). Such conditions have the highest potential to, but do not
necessarily always result in product or process failure.
Water system – A system for producing, storing and distributing water, usually compliant to a specific
pharmacopeia grade (e.g. purified water and water for injection (WFI)).
Z-value – The temperature difference that leads to a 10-fold change in the D-value of the biological
indicators.
